Deuterium and evolution of life
© Oleg Mosin, Ignat Ignatov
Oleg Mosin, PhD.,
Ignat Ignatov, DSc., Professor
Contact E-mail: mosin-oleg@yandex.ru
Introduction
The role of deuterium in molecular evolution is most interesting question of nowdays science comprises two points mainly: the evolution of deuterium itself as well as the chemical processes going with participation of deuterium. It is believed the big bang produce the universe that was much denser and hotter than it is now and made almost entirely of two main elements - hydrogen and helium. Deuterium itself was made only at a second stage of the beginning of the universe, namely through the collision of one neutron with one proton at a temperature of about one billion degrees; furthemore the two formed deuterons in turn stuck together into helium nuclei, which contain two protons and two neutrons. It is considered, that during the formation of helium nuclei, almost all the deuterons combined to form helium nuclei, leaving a tiny remant to be detected today so that only one in 10.000 deuterons remained unpaired.
Thus, deuterium serves as a particularly important marker. The quantity of deuterium in contemporary nature is approximately small and measured as no more than 0.015% (from the whole number of hydrogen atoms) and depends strongly on both the uniformity of substance and the total amount of matter formed in course of early evolution. One may suggest, that the very reliable source of producing of deuterium theoretically may to be the numerical explosions of nova stars, but deuterium itself is very readily destroyed in those stars. If it was so, perhaps this was the answer to the question why the quantity of deuterium increased slitely during the global changes of climate for worming conditions.
Previous biological experiments with D2O and structural-conformational studies with deuterated molecules, performed by us, enable to modeling conditions under which the first living forms of life might be evolved (Ignatov & Mosin, 2013a; Ignatov & Mosin, 2013b; Ignatov & Mosin, 2013c). The content of deuterium in hot mineral water may be increased due to the physical chemical processes of the deuterium accumulation. It can be presumed that primary water might contain more deuterium at early stages of evolution of first living structures, and deuterium was distributed non-uniformly in the hydrosphere and atmosphere (Ignatov & Mosin, 2012). The primary reductive atmosphere of the Earth consisted basically of gas mixture CO, H2, N2, NH3, CH4, lacked O2–O3 layer protecting the Earth surface from rigid short-wave solar radiation carrying huge energy capable to cause radiolysis and photolysis of water. The processes accompanying accumulation of deuterium in the hydrosphere are solar radiation, volcanic geothermal processes and electric discharges in the atmosphere. These natural processes could lead to the enrichment of the hydrosphere by deuterium in the form of HDO which evaporates more slowly then H2O, and condenses faster. If this is true, this is a significant fact regarding thermal stability of deuterated macromolecules in the preservation of life under thermal conditions, because chemical bonds with participation of deuterium are stronger than those ones formed of hydrogen.
The natural prevalence of deuterium makes up approximately 0.015–0.020 at.%, and depends strongly on the uniformity of substance and the total amount of matter formed in the course of early Galaxy evolution (Linsky, 2007). Constant sources of deuterium are explosions of nova stars and thermonuclear processes frequently occurring inside the stars. Probably, it could explain a known fact, why the amount of deuterium is slightly increased during the global changes of climate in warming conditions. The gravitational field of the Earth is insufficiently strong for the retaining of lighter hydrogen, and our planet is gradually losing hydrogen as a result of its dissociation into interplanetary space. Hydrogen evaporates faster than heavy deuterium, which can be collected by the hydrosphere. Therefore, as a result of this natural process of fractionation of H/D isotopes throughout the process of Earth evolution there should be an accumulation of deuterium in the hydrosphere and surface waters, while in the atmosphere and in water vapour deuterium content tends to be low. Thus, on the planet there occurs a natural process of separation of H and D isotopes, playing an essential role in the maintenance of life on the planet.
The second point regards the influence of temperature on the processes in living matter. Recent studies have shown that the most favorable for the origin of life and living matter seem to be hot alkaline mineral waters interacting with CaCO3 (Ignatov, 2010; Ignatov & Mosin, 2013d). According to the law for conservation of energy the process of self-organization of primary organic forms in water solutions may be supported by thermal energy of magma, volcanic activity and solar radiation. According to J. Szostak, the accumulation of organic compounds in open lakes is more possible compared to the ocean (Szostak, 2011). Life began near a hydrothermal vent: an underwater spout of hot water. Geothermal activity gives more opportunities for the origination of life. In 2009 A. Mulkidjanian and M. Galperin demonstrate that the cell cytoplasm contains potassium, zinc, manganese and phosphate ions, which are not particularly widespread in the sea aquatorium (Mulkidjanian & Galperin, 2009). J. Trevors and G. Pollack proposed in 2005 that the first cells on the Earth assembled in a hydrogel environment (Trevors & Pollack , 2005). Gel environments are capable of retaining water, oily hydrocarbons, solutes, and gas bubbles, and are capable of carrying out many functions, even in the absence of a membrane. Hydrocarbons are an organic compounds consisting entirely of hydrogen and carbon. The data presented in this paper show that the origination of living matter most probably occurred in hot mineral water. This occurred in ponds and hydrothermal vents in seawater or hot mineral water. An indisputable proof of this is the presence of stromatolites fossils. They lived in warm and hot water in zones of volcanic activity, which could be heated by magma and seem to be more stable than other first sea organisms (Ignatov, 2012). Either way or not, the model of deuterium evolution provides a framework for predicting the biochemical consequences of such new fascinating ideas.
In the years immediately following the discovery of deuterium, there developed a keen interest in development of methods for uniform biological enrichment of a cell with 2H that may be best achived via growing of an organism on medium with high content of 2H2O (99% 2H), which since yet resulted in a miscellany of rather confusing data (see as an example Katz J., Crespy H. L. 1972).
The main resolute conclusion that can be derived from the most competent and comprehensive of the early studies is that high concentrationsof 2H2O are incompatible with life and reproduction and furthemore could even causing even lethal effects on a cell. However, today a many cells could be adapted to 2H2O either via employing a special methods of adaptation which of them we shall describe above, or using selected (or/and resistent to 2H2O) strains of bacterial and other origin.
In this connection the main interesting question arises-what is the nature of this interesting phenomenon of biological adaptation to 2H2O and what is the role of life important macromolecules (particularly DNA, individual proteins, and/or enzymes) in this process? It is seems very likely, that during adaptation to 2H2O the structure and conformation of [U -2H]labeled macromolecules undergoing some modifications that are more useful for the working in 2H2O-conditions. Unfortunately, there are a small number of experiments carried out with fully deuterated cells, that could confirmed that during the growth on 2H2O [U-2H]labeled macromolecules with difined isotopical structures and conformations are formed, so that a discussion about the role of deuterium on the structure and the conformation of [U-2H]labeled macromolecules in course of biolodical adaptation to 2H2O is still actual through more than four decades of years after the first description of the biological consequences of hydrogen replacement by deuterium.
To further discuss the matter, we should distingueshed mainly three aspects of biological enrichment with deuterium: chemical, biological and biophysical aspects, all of them are connected in some way with the structure of [U -2H]labeled macromolecules. Theoretically, the presence of deuterium in biological systems certainly could be manifested in more or less degree by changes in the structure and the conformation of macromolecules. Nevertheless, it is important namely what precise position in macromolecule deuterium ocupied and dipending from that the primary and secondary isotopic effects are distingueshied. For example, most important for the structure of macromolecule the hydrogen (deuterium) bonds form between different parts of the macromolecule and play a major part in determining the structure of macromolecular chains and how these structures interact with the others and also with 2H2O environment. Another important weak force is created by the three-dimentional structure of water (2H2O), which tends to force hydrophobic groups of macromolecule together in order to minimize their disruptive effect on the hydrogen (deuterium)-bonded network of water (2H2O) molecules.
On the other side the screw parameters of the proton helix are changed by the presence of deuterium so that ordinary proteins dissolved in 2H2O exhibit a more stable helical structure (Tomita K., Rich A., et all., 1962). While 2H2O probably exerts a stabilizing effect upon the three-dimentional hydrogen (deuterium)-bonded helix via forming many permanent and easily exchangeable hydrogen (deuterium) bonds in macromolecule in the presence of 2H2O (as an example the following types of bonds -COO2H; -O2H; -S2H; -N2H; N2H2 et.), the presence of nonexchangeable deuterium atoms in amino acid side chains could only be synthesized de novo as the species with only covalent bonds -C2H, causes a decrease in protein stability.
These opposing effects do not cancel with the case of protein macromolecule, and fully deuteration of a protein often results in the destabilization. As for the deuteration of DNA macromolecule, today there are not reasonable considerations that such negative effect of 2H2O on the structure and function is really existiting. Nevertheless, deuterium substitution can thus be expected to modify by changes in the structure and the conformation of both [U- 2H]labeled DNA and protein, not only the reproductionl and division systems of a cell, and cytological or even mutagenical alterations of a cell, but to a greater or lesser degree of an order of a cell.
It should be noted, however, that not only these functions but also the lipid composition of cell membrane are drastically changed during deuteration. The lipid composition of deuteriated tissue culture cells has been most complitely investigated by a certain scientists (Rothblat et all., 1963, 1964). As it is reported in these articles mammalian cells grown in 30% (v/v) 2H2O contain more lipid than do control cells. THe increase in the lipids of 2H2O grown cells is due primarily to increased amounts of triglycerids and sterol esters. Radioisotope experiments indicate that the differens are due to an enhanced synthesis of lipid. Monkey kidney cells grown in 25% (v/v) 2H2O and or irradiated with X-rays likewise showed increases of lipid. The 2H2O grown cells contained more squalene, sterol esters, sterols, and neutral fat than did either the control of X-irradiated cells. Phospholipid levels were equal for all groups of cells. Thus the effects of 2H2O on lipid synthesis are qualitatively quite similar to those of radiation damade. An interisting observation that deserves further scrutiny relates to the radiation sensitivity of deuterated cells. Usually, cells grown and irradiated in 2H2O shown much less sensivity to radiation than ordinary cells suspended in water. Suspension of ordinary cells in 2H2O did not have any effect on the reduced sensitivety became apparent.
A serious alteration in cell chemistry must be reflected in the ability of the cells to divide in the presence of 2H2O and in the manner of its division. However, a many statements suggesting that 2H2O has a specific action on cell division are common since today. Probably it may be true that rapidly proliferating cells are highly sensitive to 2H2O, but that deuterium acts only to prevent cell division is unlikely.
The rabbit cells grown on medium containing the various concentrations of 2H2O shown, that 2H2O caused a reduction in cell division rate, and this effect increased as the concentration of 2H2O or duration of exposure, or both, were increased (Lavillaureix et all., 1962). With increasing concentration of 2H2O the frequency of early metaphases increased, accompanied by proportional decreases in the other phases.
It was suggested that 2H2O blocks mitosis in the prophase and the early metaphase of many cells grown in 2H2O. The blockage, however, was overcome if the initial concentration of 2H2O was not too high and the exposure time not too long. In experiments with eggs of the fresh water cichlid fish Aequidens portalegrensis, they observed that in 30% 2H2O only one-fifth of the eggs hathed and in 50% (v/v) 2H2O none did so. Segmentation in fertilized frog eggs developed normally for 24 hours in 40% (v/v) 2H2O, after which the embryos died. It was also found by Tumanyan and Shnol that 2H2O disturbed embryogenesis in Drosophila melanogaster eggs (Lavillaureix et all., 1962. Feeding female flies with 20% (v/v) 2H2O caused a significant increase in the proportion of nondeveloped eggs, whether males were deuterated or not.
As pointed out by many researches, carried elsewhere, the reason for the cessation of mitotic activity from exposure to 2H2O is not clear. Certain microorganisms have been adapted to grow on fully deuterated media. However, higher plants and animals resist adaptation to 2H2O. Even in microorganisms, however, cell division appears initially to be strongly inhibited upon transfer to highly deuterated media.
After the adaptation, however, cellular proliferation proceeds more or less normally in 2H2O, but this stage is not reached in higher organisms. No ready explanation in terms of the present understanding of mitosis suggests itself. In Arbacia eggs antimitotic action of 2H2O is manifested almost immediately at all stages of the mitotic cycle and during cytokinesis (Gross P. R., et all., 1963, 1964).
A stabilizing action on the nuclear membrane and gel structures, i.e., aster, spindle, and peripheral plasmagel layer of the cytoplasm, can be detected. Prophase and metaphase cells in 80% (v/v) 2H2O remain frozen in the initial state for at least 30 minutes. Furrowing capacity probably is not abolished by 2H2O. The 2H2O-block is released on immersion in 2H2O although cells kept in deuterium-rich media for long periods show multipolar and irregular divisions after removal to 2H2O, and may subsequently cytolyze. The inhibition of mitosis in the fertilized egg is not the only interesting effect of deuterium. The unfertilized egg also responds. It was described by Gross that deuterium parthenogenesis in Arbacia in the following graphic terms: if an unfertilized egg is placed in 2H2O, there appear in the cytoplasm, after half an hour, a number of cytasters. The number then increases with time. If, after an hours immersion in 2H2O, eggs are transferred to normal sea water, a high proportion (80% of the population) raises a fertilization membrane, which gives evidence that activation has occurred.
Deuterium genetics is, for the most part, like genetics itself, conveniently divisible into dipteran mutation studies, the genetics of microorganisms, and miscellaneous studies of which those of Gross and Harding, and Flaumenhaft et al. are examples. The customary procedure in most of the dipteran and bacterial investigations so far reported has been to administer 2H2O to the organism and then to test it for mutation or other chromosomal change. The results obtained by such an investigation have seldom been striking. For example, many researchers found an increase in sex-linked lethals in the sperm of flies that had been exposed to deuterium, either by way of injection into their pupae, or by the inclusion of 2H2O in their food. They introduced 2H2O into Drosophila melanogaster larvae both by feeding and by injection. The males which matured from these larvae were tested for mutation by CIB method. But the test showed no increase in the mutation rate. It was assumed by these scientists that the deuterium which was used in dilute form entered the DNA molecule.
De Giovanni and Zamenhof have carried out the most comprehensive investigations on the genetic effects of deuterium in bacteria. The results are of considerable interest. For example, they found a several mutants of E. coli, including a so called rough mutant 1/D which is more resistant to 2H2O than its parent strain, were isolated from E. coli grown in 2H2O media. The spontaneous frequency of occurerence of this mutant was 10-4, and the mutation rate could be increased 300-fold by ultraviolet irradiation. This mutant was derived only from the strain E. coli 15 thymidine, and no similar mutant was observed in other strains of E. coli or B. subtilis. By application of a fluctuation test, De Giovanni then was able to show convincingly that this mutation to increased deuterium resistance occurred spontaneously and not in response to the mutagenic effect of 2H2O. Back mutations in some instances do seem to occur at higher rates in 2H2O. Reversion from streptomycin dependence to streptomycin sensitivity in E. coli strain Sd/4, or from thymine dependence to thymine independence in strain 1 occurs with higher frequency in 2H2O, but 2H2O does not cause a discernible increase in mutation in the wild type.
De Giovanni further found that deuteriated purines and pryrimidines had no effect upon the growth and back mutation rates of specific base-requiring strains. Thymine containing deuterium in two of the four nonexchangeable positions adequately supplied the requirement for thymine with no concominant genetic changes. It would appear therefore that the preponderance of the evidence from these studies with bacteria is in favor of the view that 2H2O is not a strong mutagenic agent.
It was reported by many researchers a series experiments designed to test the ability of deuterium to produce mutation and nondisjunction. Deuterium like tritium appears to increase non-disjunction, but either agent separately is less effective than the two acting together. Hughes and Hildreth exposed male flies which had been grown on a 20% (v/v) 2H2O diet to an irradiation of 1000 r. of X-rays. It was found that there was not significant difference in the frequency of observed mutations between 2H2O flies and normal flies subjected to the same radiation.
Tumanyan and Shnol also found no mutagenic effect of 2H2O on recessive and dominant lethal marks in D. melanogaster, inbred line Domodedovo 18. Flaumenhaft and Katz grew fully deuteriated E. coli in 99,6% (v/v) 2H2O with fully deuteriated substrates, and found that the mutation rate after ultraviolet irradiation was distinctly lower than that of nondeuteriated organisms. The simultaneous presence of both deuterium and protium in nearly equal proportions in the constituent molecule of an organism could conceivably create difficulties for the organism since the rate pattern would be seriously distorted. They further found that cells grown in 2H2O and then transferred to 2H2O showed an enhanced susceptibility to ultraviolet irradiation. This suggests that organisms containing both hydrogen or deuterium, but it leaves unanswered the question of why serial subculture in H2O-2H2O media is required for adaptation of many organisms.
Many researchers studied the growth of phage T4 in E. coli cells which were cultivated in media containing various concentrations of 2H2O from zero to 95% (v/v). No significant increase in forward mutation in this phage could be observed, but the rate for reverse mutation was increased, and reached a maximum in phage grown in 50% (v/v) 2H2O. Although it was reported that a further increase in H2O concentration up to 90% (v/v) producers little augmentation of the reversion index, the actual data presented by Konrad indicates a decided increase in reverse mutation rate in phage exposed to more than 50% (v/v) 2H2O.
There have been carried out a big deal of cytochemical study of fully deuteriated microorganisms grown autotrophically for very long periods in 2H2O (Flaumenhaft E., Conrad S. M., and Katz J. J., 1960a, 1960b). The main conclusion that could be made from these studies is that the nucleus of deuterated cells was much larger than that of nondeuterated cells, and it contained greater amounts of DNA. Also present were much greater amounts of rather widely scattered cytoplasmic RNA within the cells. It was found also, that deuterated cells stained much more darkly for proteins, indicating higher concentrations of free basic groups. Both fluorescence and electron microscopy indicated that deuteration results in readily observable morphological changes. For example, the chloroplast structure of deuteriated plants organisms was more primitive in appearance, less well-differentiated, and distinctly less well-organized. The very interesting conclusion was made, then a low or/and high temperature grown organisms implied the morphological consequences of extensive isotopic replacement of hydrogen by deuterium so that in some respects resemble with the effects produced by reduction or/and increase in temperature of growth.
But, paradoxically as shown numerious studies on biological adaptation to 2H2O, a many cells of bacterial and algae origin could, nevertheless, well grown on absolute 2H2O and, therefore, to stabilize their biological apparatus and the structure of macromolecules for working in the presence of 2H2O. The mechanism of this stabilization nor at a level of the structure of [U-2H]labeled macromolecules or at a level of their functional properties is not yet complitely understood. We still don’t know what possibilities a cell used for adaptation to 2H2O. We can only say, that probably, it a complex phenomenon resulting both from the changes in structural and the physiological level of a macrosystem. That is why there is every prospect that continued investigation of deuterium isotope effects in living organisms will yield results of both scientific and practical importance, for it is precisely. For example, the studies of the structure and the functioning of biolodical important [U -2H]labeled macromolecules obtained via biological adaptaition to high concentrations of 2H2O are most attract an attention of medical scientists as a simple way for creating a fully deuterated forms of DNA and special enzymes could well be working in a certain biotechnological processes required the presence of 2H2O. Secondly, if the structure of fully deuterated proteins may be stabilized in 2H2O in a view of duarability of deuterated bonds, it would be very interesting to study the thermo-stability of [U -2H]labeled proteins for using them directly in processes going at high temperatures.
It would be very perspective if someone could create the thermo-stable proteins simply via deuteration of the macromolecules by growing a cell-producent on 2H2O wit 99% 2H. Third, particular interest have also the studies on the role of primodial deuterium in molecular evolution. The solution of these obscure questions concerning the biological adaptation to 2H2O should cast a new light on molecular evolution in a view of the preferable selection of macromolecules with difined deuterated structures. Thus, the main purpose of the present project is the studies of the structure and the function of fully deuterated macromolecules (particularly DNA and individual proteins and/or enzymes) obtained via biological adaptation to high concentrations of 2H2O.
To carry out the studies with fully deuterated macromolecules one must firstly to obtain the appropriate deuterated material with high level of enrichment for isolation of pure DNA and individual proteins to which the various methods of stable isotope detection further can be applyed. For example, the three-dimentional NMR combined together with the method of X-ray diffraction, infrared (IR)-, laser spectrometry and circular dichroism (CD) is a well proved method for the studies of the structure and the functioning of [U -2H]labeled macromolecules, and for investigations of various aspects of their biophysical behavior. Taking into account the ecological aspect of using [U -2H]labeled compounds, it should be noted in conclusion, that the preferable properties of applying deuterium for biochemical studies are caused mainly by the absence of radioactivity of deuterium that is the most important fact for carrying out the biological incorporation of deuterium into organism.
The methods for analyzing the structure and the conformation
of [U -2H]labeled macromolecules.
The biological labelling with deuterium is a useful tool for investigating the structure and the conformational properties of macromolecules. The fundamental objectives have meant that living models have retained their importance for functional studies of such biological important macromolecules and can be used to obtain structural and dynamic information about the [U -2H]labeled macromolecules.
The method of X-ray diffraction should be noted as a indespencible tool for determing the details of the three-dimentional structure of globular proteins and other macromolecules (Mathews C. K., van Holde K. E., 1996). Yet this technique has the fundamental limitation that it can be employed only when the molecules are crystallized, and crystallization is not always easy or even possible. Furthermore, this method cannot easily be used to study the conformational changes in response to changes in the molecules environment.
Other methods, for example IR-spectroscopy, can provide direct information concerning the macromolecular structure. For example, the exact positions of infrared bands corresponding to vibrations in the polypeptide backbone are sensitive to the conformational state (a helix, b sheet et.) of the chain (Campbell I. D., and Dwek R. A., 1984). Thus, the studies in this region of the spectrum are often used to investigate the conformations of protein molecules.
Although, IR-, and absorption spectroscopy can be helpful in following molecular changes, such measurements are difficult to interpret directly in terms of changes of secondary structure. For this purpose, techniques of circular dichroism involving polarized light have become important (Johnson W. C., 1990). For example, if a protein is denatured so that its native structure, containing a helix and b sheet regions, is transformed into an unfolded, random-coil structure, this transformation will be reflected in a dramatic change in its CD spectrum. Circular dichroism can be used in another way, to estimate the content of a helix and b sheet in native proteins. The contributions of these different secondary structures to their circular dichroism at different wavelenghths are known, so we may attempt to match an observed spectrum of protein by a combination of such contributions.
Although circular dichroism is an extremely useful technique, it is not a very discriminating one. That is, it cannot, at present, tell us what is happening at a particular point in a protein molecule. A method that has the great potential to do so is nuclear magnetic resonance. This advance now makes it possible to use NMR to study big varieties of DNA and proteins with more complex biological functions functioning in natural liquid environment. Often these proteins have more than one domain and more than one site of interaction. Allosteric systems, receptors and small molecule ligand-modulated DNA-binding proteins and DNA are some examples of the molecular systems which can now be analysed in molecular detail. For example, due to the development of two-dimentional Fourier transformation techniques, NMR spectroscopy has become a powerful tool for determining the protein structure and conformation (Fesic S. W. and Zuiderweg E. R., 1990).
The preparation of [U- 2H]labeled macromolecules.
Through technical advances of biotechnology, many macromolecules, for example a certain individual proteins are successfuly cloned and can be obtained in large quantities by expression in microbial and/or mammalian systems, so that an ever-increasing number of individual [U- 2H]labeled macromolecules from various biological objects are becoming commercially available. It should be noted, however, that the application of various methods for the preparation of [U -2H]labeled macromolecules (chemical or biosynthetical) often results in obtaining the forms of molecules with different number of protons substituted by deuterium, the phenomenon that is known as heterogenious labelling, so that the special methods for the preparation of [U -2H]labeled macromolecules should be applyed to minimaze this process. For example, the proteins containing only deuterium atoms in polypeptide chain of macromolecule can be produced biotechnologically with using the special genetically constructed strains of bacteria carrying the mutations of geens excluding the metabolic exchange between the parterns of unlabeled intermediators during the biosynthesis of [U -2H]labeled macromolecules.
I may briefly indicate three possibilities for deuterium enrichment:
(1) to grow the organism on a minium salt medium with content of 2H2O 99% 2H;
(2) To grow the organism on a medium supplemented with 99% 2H2O and [U -2H]labeled amino acid mixture.
(3) the isotopic exchange of susceptible protons in amino acid residues already incorporated into protein.
Method 1 is very useful for the preparation of [U- 2H]labeled macromolecules if only applyed strains of bacterial or different origin could well be grown on minimal media in the presence of high concentrations of 2H2O. Very often in this case the biological adaptation to 2HO is required. Method 2, while generally applicable, is limited by the difficulty and expense of preparing fully deuterated amino acid mixtures from algae grown on 2H2O. However, recently we proposed to use a fully deuterated biomass of methlotrophic bacterium B. methylicum with protein content about 55% (from dry weight) obtained via multistep adaptaition to 98% (v/v) 2H2O and 2% (v/v) [U-2H]MetOH as growth substrates for growing the other bacterial strains to prepare a gram quantities of [U -2H]labeled amino acids, proteins and nucleosites with high levels of enrichment (90.0-97.5% 2H) (Mosin O. V., Karnaukhova E. N., Pshenichnikova A. B.; 1994; Skladnev D. A., Mosin O. V., et all; 1996; Shvets V. I., Yurkevich A. M., Mosin O. V.; 1995).
Method 2 is also necessary when the organism will not grow on a minimal medium as it was in the case with the applying the bacteria requiring the complex composition media for their growth. This approach will also be necessary for the labeling of proteins expressed in systems other than E. coli (e.g. yeast, insect, and mammalian expression systems) which may be important for the proper folding of proteins from higher organisms. Since the protons of interest in proteins are most often carbon bound and thus do not exchange under mild conditions, method 3 is severely limited by stability of proteins under the harsh conditions necessary for (1H-2H) exchange.
The main hypothesis.
We proposed that a cell theoretically could in principle synthezise a big number of forms of [2H]labeled macromolecules with somewhat different structures and conformations, so that a cell could easily select a preferable one from al these species in a course of adaptation to 2H2O, that is the best suitable namely for that conditions. A simple imaginary principle I am going to discuss here perhaps somewhat may explain this probable mechanism. Let us suppose, for example that there are at least two imadinary structural systems - ordinary (normal) system call it a system 1 and unordinary (adaptive) system 2 (see a Figure above). Supporse, that the environment is a homoginious substanse and compose from ordinary substance A (H2O) (situation 1). The necessarely condition for the normal working of this model in natural H2O environment is that system 1 works and system 2 stay in background (situation 2). Suppose that the environment have changed for substance B (2H2O). Then the system 2 will work, while the system 1 will stay in background (situation 2). When environment will be the natural again, the system 1 will begin the work again, while the system 2 will stay in background. Admit that the two systems both presented at the time being and could be regulated in such way that they may switch bitween each other during the working so that the model system does not undergoing the considerable alterations.
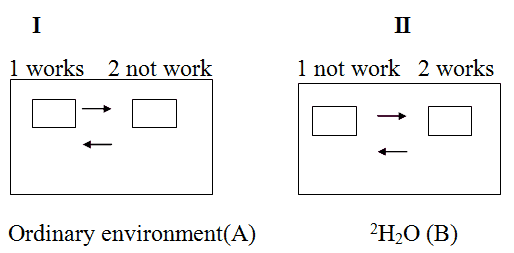
Fig. 1. The imaginary principle of realization of biological adaptation
Phenomenon of biological adaptation to 2H2O.
Our research has confirmed, that ability to adaptation to 2Н2О is differed for various species of bacteria and can to be varried even in frames of one taxonomic family (Mosin O. V. et al., 1996a, 1996b).From this, it is possible to conclude, that the adaptation to 2Н2О is determined both by taxonomic specifity of the organism, and peculiarities of the metabolism, as well as by functioning of various ways of accimilation of hydrogen (deuterium) substrates, as well as evolutionary level, which an object itself occupies. The less a level of evolutionary development of an organism, the better it therefore adapts itself to 2H2O. For example, there are halophilic bacteria that are being the most primitive in the evolutionary plan, and therefore, they practically not requiring carry out a special adaptation methods to grow on 2Н2О. On the contrary, bacills (eubacteria) and methylotrophs (gram-negative bacteria) worse adapted to 2Н2О.
At the same time for all tested cells the growth on 2H2O was accompanied by considerable decrease of a level of biosynthesis of appropriated cellular compounds. The data obtained confirm that the adaptation to 2Н2О is a rather phenotypical phenomenon, as the adapted cells could be returned to a normal growth and biosynthesis in protonated media after lag-phase (Mosin O. V. et al., 1993).
However, when the adaptive process goes continuously during the many generation, the population of cells can use a special genetic mechanisms for the adaptation to 2H2O. For example, mutations of geens can be resulted in amino acid replacements in molecules of proteins, which in turn could cause a formation of new isoenzymes, and in the special cases - even the anomal working enzymes of a newer structure type. The replacements of these compounds can ensure a development of new ways of regulation of enzymic activity, ensuring more adequate reaction to signals, causing possible changes in speeds and specifity of metabolic processes.
Despite it, the basic reactions of metabolism of adapted cells probably do not undergo essential changes in 2Н2О. At the same time the effect of convertibility of growth on Н2О/2Н2О - does not theoretically exclude an opportunity that this attribute is stably kept when cells grown on 2Н2О, but masks when transfer the cells on deuterated medium.
However, here it is necessary to emphasize, that for realization of biological adaptation to 2H2O the composition of growth medium plays an important role. In this case it is not excluded, that during the adaptation on the minimal medium, containing 2Н2О there are formed the forms of bacteria, auxotrophic on a certain growth factors (for example amino acids et) and thereof bacterial growth is inhibited while grown on these media. At the same time the adaptation to 2Н2О occurs best on complex media, the composition of which coul compensate the requirement in those growth factors.
It is possible also to assume, that the macromolecules realize the special mechanisms, which promote a stabilization of their structure in 2H2O and the functional reorganization for best working in 2Н2О. Thus, the distinctions in nuclear mass of hydrogen atom and deuterium can indirectly to be a reason of distinctions in synthesis of deuterated forms of DNA and proteins, which can be resulting in the structural distinctions and, hence, to functional changes in [2H]labeled macromolecules. Hawever, it is not excluded, that during incubation on 2Н2О the enzymes do not stop the function, but changes stipulating by isotopic replacement due to the primary and secondary isotopic effects as well as by the action of 2Н2О as solvent (density, viscosity) in comparison with Н2О are resulted in changes of speeds and specifics of metabolic reactions.
In the case with biological adaptation to 2H2O we should inspect the following types of adaptive mechanisms:
1. adaptation at a level of macromolecular components of cells: It is possible to allocate mainly two kinds of such adaptation:
(a). Differences of intracellular concentration of macromolecules;
(b). The forming in 2H2O the deuterated macromolecules with other conformations, which could be replaced the ordinary protonated macromolecules synthesized by cells in normal conditions.
We suppose, that in principle, any protein macromolecule could adopt an almost unlimited number of conformations. Most pilypeptide chains, however, fold into only one particular conformation determined by their amino acid sequence. That is because the side chains of the amino acids associate with one another and with water (2H2O) to form various weak noncovalent bonds. Provided that the appropriate side chains are present at crucial positions in the chain, large forces are developed that make one particular conformation especially stable.
These two strategies of adaptation could possible to be distinqueshed accordinly as "quantitative" and "qualitative" strategies;
2. adaptation at a level of microenvironment in wich macromolecules are submerged: the essence of this mechanism is, that the adaptive change of structural and conformational properties of [2H]labeled macromolecules is conditioned both by directional action of 2H2O environment on a growth of cells and by its physico-chemical structure (osmotic pressure, viscosity, density, рН, concentration of 2H2O).
2H2O appeared to stabilize the plasmagel structure of biological microenvironment. The external pressure required to make the cells assume a spherical shape increased 3.6 kg/cm2 for each per cent increase in the presence of 2H2O. It thus seems well established that deuteration can affect the mechanical properties of cytoplasm, and that this factor must be taken into account in assessing the consequences of isotopic substitution of macromolecules. In model experiments with gelatin structure, it was demonstrated that in 2H2O there is a greater protein-protein interaction than in H2O (Scheraga J. A; 1960).
A progressive increase in the melting temperature of the gel in 2H2O is observed accompanied by an increase in the reduced viscosity. That 2H2O can have marked effects on the physical properties of proteins has been known for some time. Consequently it is natural to attribute changes in the mechanical properties of cell structures induced by 2H2O to protein response. Nevertheless, the effects of deuterium on proteins, while real, must be only a partial explanation of the situation. The interaction of proteins with solvent water is extraordinarily complex, and the exact nature of the protein is crucial in determining the magnitude of changes resulting from the replacement of H2O by 2H2O.
This mechanism has extremely large importance and supplements the macromolecular adaptation; 3. adaptation at a functional level, when the change of an overall performance of macromolecular systems, is not connected with a change of a number of macromolecules being available or with the macromolecules of their types. Adaptation in this case could provide the changes by using the already existing macromolecular systems - according to requirements by this or that metabolic activity.
Table 1. Some physical constants of H2O and 2H2O
Physical constant
H2O
2Н2О
Density, d20 (g/c.c)
0,9982
1,1056
Molecular volume, V20 (ml/mole)
18,05
18,12
Viscosity m20 (centipose)
1,005
1,25
Melting point (0C)
0,1
3,82
Boiling point (0C)
100,0
101,72
Temperature of maximum density (0C)
4,0
11,6
Ion product (25 0C)
10-14
0,3x10-14
Heat of formation (cal/mole)
-68,318
-70,414
Free energy of formation (cal/mole)
-56,693
-58,201
Entropy (e.u/mole)
45,14
47,41
Secondary effects may still be of importance in biological systems sensitive to kinetic distortions. Deuterium also affects equilibrium constants, particularly the ionization constants of weak acids and bases in composition of macromolecules dissolved in heavy water (see a Table below). Acid strength of macromolecules in 2H2O is decreased by factors of 2 to 5, and consequently, the rates of acid-base catalyzed reactions may be greatly different in 2H2O as compared to H2O. Such reactions frequently may be a faster in 2H2O than H2O solution (Covington A. K., Robinson R. A., and Bates R. G., 1966; Glasoe P. K., and Long F. A., 1960).
The natural water consists on 99.7 mol.% of H216O, which molecules are formed by 1H and 16O atoms. The remaining 0.3 mol.% is represented by isotope varieties (isotopologues) of water molecules, wherein deuterium forms 6 configurations of isotopologues – HD16O, HD17O, HD18O, D216O, D217O, D218O, while 3 configuration are formed by isotopologues of oxygen – Н216O, Н217O, Н218O.
The average ratio of atoms of deuterium and hydrogen in natural waters compiles ~1:5700. In natural waters, the deuterium content is distributed irregularly: from 0.02–0.03 mol.% for river and sea water, to 0.015 mol.% for water of Antarctic ice – the most purified from deuterium natural water containing deuterium in 1.5 times less than that of seawater.
The concentration of water molecules containing heavy isotopes of D, 17O and 18O, in natural water varies within the limits laid down in the basic standards of the isotopic composition of the hydrosphere SNOW and SLAP (Table 2).
According to the international SMOW standard the absolute content of D (isotopic shift, δ, ppm) in sea water: D/H = (155.76±0.05).10-6 (155.76 ppm) (Ignatov & Mosin, 2013d). For SLAP standard isotopic shifts for D in seawater: D/H = 89.10-6 (89 ppm). Content of the lightest isotopologue – H216O in water corresponding to SMOW standard is 997.0325 g/kg (99.73 mol.%), and for SLAP standard – 997.3179 g/kg (99.76 mol.%). In surface waters, the ratio D/H = (1.32–1.51).10-4, while in the coastal seawater – (1.55–1.56).10-4.
The natural waters of CIS countries are characterized by negative deviations from SMOW standard to (1.0–1.5).10-5, in some places up to (6.0–6.7).10-5, but there are also observed positive deviations at 2.0.10-5. Waters of other underground and surface water sources contain varied amounts of deuterium (isotopic shifts) – from δ = +5.0 D,%, SMOW (Mediterranean Sea) up to δ = -105 D,%, SMOW (Volga River).
Table 2. The calculated mass concentrations of isotopologues in natural water corresponding to international standards of SMOW* and SLAP**
- Isotopologue; Molecular mass, u; Isotopic content, g/kg
- SMOW; SLAP
- 1H216O; 18.01056470; 997.032536356; 997.317982662
- 1HD16O; 19.01684144; 0.328000097; 0.187668379
- D216O; 20.02311819; 0.000026900; 0.000008804
- 1H217O; 19.01478127; 0.411509070; 0.388988825
- 1HD17O; 20.02105801; 0.000134998; 0.000072993
- D217O; 21.02733476; 0.000000011; 0.000000003
- 1H218O; 20.01481037; 2.227063738; 2.104884332
- 1HD18O; 21.02108711; 0.000728769; 0.000393984
- D218O; 22.02736386; 0.000000059; 0.000000018
Notes:
*SMOW (average molecular weight = 18.01528873 u)
**SLAP (average molecular weight = 18.01491202 u)
The equilibrium vapor pressure of water isotopologues is differed quite significantly. The smaller the mass of the water molecule, the higher the vapor pressure, meaning that the vapor being in equilibrium with water, is always enriched with light isotopes of oxygen and hydrogen. Due to relatively low-mass of elements the difference between the mass of isotopes is large, therefore they are fractionated in natural processes: D/H → 100 %, 18O/16O → 12.5 %. Isotopes of hydrogen and oxygen are more efficiently fractionated via the processes of evaporation-condensation and water crystallization. The isotopic fractionation is carried out by following methods – isotopic exchange in the presence of Pd and Pt, the electrolysis of water in combination with a catalytic isotopic exchange between H2O and H2, column rectification of cooled gaseous H2, vacuum freezing of cold vapor followed by thawing and other (Mosin, 2012).
The chemical isotopic effect of 2H2O.
The effect of isotopic replacement that has particularly attracted the attention of chemists is the kinetic isotope effect (Thomson J. F., 1963). The substitution of deuterium for hydrogen in a chemical bond of macromolecules can markedly affect the rate of scission of this bond, and so exert pronounced effects on the relative rates of chemical reactions going in 2H2O with participation of macromolecules. This change in rate of scission of a bond resulting from the substitution of deuterium for hydrogen is a primary isotopic effect. The direction and magnitude of the isotope effect will depend on the kind of transition state involved in the activated reaction complex, but in general, deuterium depresses reaction rates. The usual terminology of the chemist to describe the primary kinetic effect is in terms of the ratio of the specific rate constants kh/kd. The maximum positive primary kinetic isotopic effect which can be expected at ordinary temperatures in a chemical reaction leading to rupture of bonds involving hydrogen can be readily calculated, and the maximum ratio kh/kd in macromolecules is in the range of 7 to 10 for C-H versus C-2H, N-H versus N-2H, and O-H versus O-2H bonds. However, maximum ratios are seldom observed for a variety of reasons, but values of kh/kd in the range of 2 to 5 are common (Wiberg K. B., 1955). Deuterium located at positions in a macromolecule other than at the reaction locus can also affect the rate of a reaction. Such an effect is a secondary isotope effect and is usually much smaller than a primary isotope effect.
In general, when the macromolecules transfer to deuterated medium not only water due to the reaction of an exchange (Н2О -2Н2О) dilutes with deuterium, but also occurs a very fast isotopic (1Н-2Н)-exchange in hydroxylic (-OH), carboxilic (-COOH), sulfurhydrilic (-SH) and nitrogen (-NH; -NH2) groups of all organic compounds including the nucleic acids and proteins. It is known, that in these conditions only С-2Н bond is not exposed to isotopic exchange and thereof only the species of macromolecules with С-2H type of bonds can be synthesized de novo. This is very probably, that the most effects, observed at adaptation to 2Н2О are connected with the formation in 2Н2О [U -2H]labeled molecules with conformations having the other structural and dynamic properties, than conformations, formed with participation of hydrogen, and consequently having other activity and biophysical properties.
So, according to the theory of absolute speeds the break of С-1H-bonds can occur faster, than С-2H-bonds (C-2H-bonds are more durable than C-1 , mobility of an ion 2H+ is less, than mobility of 1Н+, the constant of ionization 2Н2О is a little bit less than ionization constant of 2Н2О. Thus, in principle, the structures of [U -2H]labeled macromolecules may to be more friable that those are forming in ordinary H2O. But, nevertheless, the stability of [U -2H]labeled macromolecules probably depending on what particular bond is labeled with deuterium (covalent bonds -C2H that causing the instability or hydrogen bonds causing the stabilization of conformation of macromolecules via forming the three-dimentional netwok of hydrogen(deuterum) bonds in macromolecule) and what precise position of the macromolecule was labeled with deuterium. For example, the very valuable and sensitive for deuterium substitution position in macromolecule is the reactive center (primary isotopic effects). The non-essential positions in macromolecule are those ones that situated far away from the reactive center of macromolecule (secondary isotopic effects). It is also possible to make a conclusion, that the sensitivity of various macromolecules to substitution on 2Н bears the individual character and depending on the structure of macromolecule itself, and thus, can be varried. From the point of view of physical chemistry, the most sensitive to replacement of 1Н+ on 2H+ can appear the apparatus of macromolecular biosyntesis and respiration system, those ones, which use high mobility of protons (deuterons) and high speed of break of hydrogen (deuterium) bonds. From that it is posible to assume, that the macromolecules should realize a special mechanisms (both at a level of primary structure and a folding of macromolecules) which could promote the stabilizition of the macromolecular structure in 2H2O and somewhat the functional reorganization of their work in 2H2O.
A principal feature of the structure of such biologically important compounds as proteins and nucleic acids is the maintenance of their structure by virtue of the participation of many hydrogen bonds in macromolecule. One may expect that the hydrogen bonds formed by of many deuterium will be different in their energy from those formed by proton. The differences in the nuclear mass of hydrogen and deuterium may possibly cause disturbances in the DNA-synthesis, leading to permanent changes in its structure and consequently in the cells genotype. The multiplication which would occur in macromolecules of even a small difference between a proton and a deuteron bond would certainly have the effect upon its structure.
The sensitivity of enzyme function to structure and the presumed sensitivity of nucleic acids function (genetic and mitotic) to its structure would lead one to expect a noticeable effect on the metabolic pattern and reproductive behavior of the organism. And next, the changes in dissociation constants of DNA and protein ionizable groups when transfer the macromolecule from water to 2H2O may perturb the charge state of the DNA and protein. Substitution of 1H for deuterium also affects the stability and geometry of hydrogen bonds in apparently rather complex way and may, through the changes in the hydrogen bond zero-point vibrational energies, alter the conformational dynamics of hydrogen (deuterium)-bonded structures within the DNA and protein in 2H2O.
Numerous studies carried out by us with various biological objects in 2H2O, proved that when biological objects being exposed to water with different deuterium content, their reaction varies depending on the isotopic composition of water (the content of deuterium in water) and magnitude of isotope effects determined by the difference of constants of chemical reactions rates kH/kD in H2O and 2H2O. The maximum kinetic isotopic effect observed at ordinary temperatures in chemical reactions leading to rupture of bonds involving hydrogen and deuterium atoms lies in the range kH/kD = 5–8 for C–H versus C–2H, N–2H versus N–2H, and O–2H versus O–2H-bonds (Mosin, 1996; Mosin & Ignatov, 2012a; Mosin & Ignatov, 2012b). Isotopic effects have an impact not only on the physical and chemical properties of deuterated macromolecules in which H atoms are substituted with 2H atoms, but also on the biological behaviour of biological objects in 2H2O. Experiments with 2H2O (Table 1) have shown, that green-blue algae is capable to grow on 70 % (v/v) 2H2O, methylotrophic bacteria – 75 % (v/v) 2H2O, chemoheterotrophic bacteria – 82 % (v/v) 2H2O, and photo-organotrophic halobacteria – 95 % (v/v) 2H2O.
Our studies indicated that the ability of adaptation to 2Н2О for different taxonomic groups of microorganisms is different, and stipulated by taxonomic affiliation, metabolic characteristics, pathways of assimilation of substrates, as well as by evolutionary niche occupied by the object. Thus, the lower the level of evolutionary organization of the organism, the easier it adapted to the presence of deuterium in growth media. Thus, most primitive in evolutionary terms (cell membrane structure, cell organization, resistance to environmental factors) of the studied objects are photo-organotrophic halobacteria related to archaebacteria, standing apart from both prokaryotic and eukaryotic microorganisms, exhibiting increased resistance to 2Н2О and practically needed no adaptation to 2Н2О, contrary to blue-green algae, which, being eukaryotes, are the more difficult adapted to 2Н2О and, therefore, exhibit inhibition of growth at 70–75 % (v/v) 2H2О.
The composition of growth media evidently also plays an important role in process of adaptation to 2Н2О, because the reason of inhibition of cell growth and cell death can be changes of the parity ratio of synthesized metabolites in 2Н2О-media: amino acids, proteins and carbohydrates. It is noted that adaptation to 2Н2О occures easier on complex growth media than on the minimal growth media with full substrates at a gradual increasing of deuterium content in the growth media, as the sensitivity to 2Н2О of different vital systems is different. As a rule, even highly deuterated growth media contain remaining protons ~0,2–10 atom.%. These remaining protons facilitate the restructuring to the changed conditions during the adaptation to 2Н2О, presumably integrating into those sites, which are the most sensitive to the replacement of hydrogen by deuterium. The evidence has been obtained that cells evidently are able to regulate the 2Н/1H ratios, while its changes trigger distinct molecular processes. One possibility to modify intracellular 2Н/1H ratios is the activation of the H+-transport system, which preferentially eliminates H+, resulting in increased 2Н/1H ratios within cells (Somlyai et al., 2012). Furthermore deuterium induces physiological, morphological and cytological alterations on the cell. There were marked the significant differences in the morphology of the protonated and deuterated cells of blue-green algae C. vulgaris. Cells grown on 2Н2О-media were ~2–3 times larger in size and had thicker cell walls, than the control cells grown on a conventional protonated growth media with ordinary water, the distribution of DNA in them was non-uniform. In some cases on on the surface of cell membranes may be observed areas consisting of tightly packed pleats of a cytoplasmic membrane resembling mezosoms – intracytoplasmic bacterial membrane of vesicular structure and tubular form formed by the invasion of cytoplasmic membrane into the cytoplasm (Figure 10). It is assumed that mezosoms involved in the formation of cell walls, replication and segregation of DNA, nucleotides and other processes. There is also evidence that the majority number of mezosoms being absent in normal cells is formed by a chemical action of some external factors – low and high temperatures, fluctuation of pH and and other factors. Furthermore, deuterated cells of C. vulgaris were also characterized by a drastic change in cell form and direction of their division. The observed cell division cytodieresis did not end by the usual divergence of the daughter cells, but led to the formation of abnormal cells, as described by other authors (Eryomin et al., 1978). The observed morphological changes associated with the inhibition of growth of deuterated cells were stipulated by the cell restructuring during the process of adaptation to 2Н2О. The fact that the deuterated cells are larger in size (apparent size was of ~2–4 times larger than the size of the protonated cells), apparently is a general biological phenomenn proved by growing a number of other adapted to 2Н2О prokaryotic and eukaryotic cells (Mosin & Ignatov, 2012a; Mosin & Ignatov, 2012b; Mosin & Ignatov, 2014).
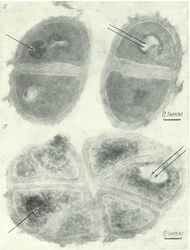
Fig. 2. Electron micrographs of Micrococcus lysodeikticus cells obtained by SEM method: a) – protonated cells obtained on H2O-medium; b) – deuterated cells obtained on 2Н2О-medium. The arrows indicate the tightly-packed portions of the membranesOur data generally confirm a stable notion that adaptation to 2Н2О is a phenotypic phenomenon as the adapted cells eventually return back to the normal growth after some lag-period after their replacement back onto H2O-medium. However, the effect of reversion of growth on H2O/2Н2О media does not exclude an opportunity that a certain genotype determines the manifistation of the same phenotypic attribute in 2Н2О-media with high deuterium content. At placing a cell onto 2Н2О-media lacking protons, not only 2Н2О is removed from a cell due to isotopic (1H–2Н) exchange, but also there are occurred a rapid isotopic (1H–2Н) exchange in hydroxyl (-OH), sulfohydryl (-SH) and amino (-NH2) groups in all molecules of organic substances, including proteins, nucleic acids, carbohydrates and lipids. It is known, that in these conditions only covalent C–H bond is not exposed to isotopic (1H–2Н) exchange and, thereof only molecules with bonds such as C–2Н can be synthesized de novo (Mosin et al., 1996b; Mosin & Ignatov, 2012a). Depending on the position of the deuterium atom in the molecule, there are distinguished primary and secondary isotopic effects mediated by intermolecular interactions. In this aspect, the most important for the structure of macromolecules are dynamic short-lived hydrogen (deuterium) bonds formed between the electron deficient 1H(2Н) atoms and adjacent electronegative O, C, N, S- heteroatoms in the molecules, acting as acceptors of H-bond (Ignatov & Mosin, 2013c). The hydrogen bond, based on weak electrostatic forces, donor-acceptor interactions with charge-transfer and intermolecular van der Waals forces, is of the vital importance in the chemistry of intermolecular interactions and maintaining the spatial structure of macromolecules in aqueous solutions (Ignatov & Mosin, 2013d). Another important property is defined by the three-dimensional structure of 2Н2О molecule having the tendency to pull together hydrophobic groups of macromolecules to minimize their disruptive effect on the hydrogen (deuterium)-bonded network in 2Н2О. This leads to stabilization of the structure of protein and nucleic acid macromolecules in the presence of 2Н2О. That is why, the structure of macromolecules of proteins and nucleic acids in the presence of 2Н2О, is somehow stabilized (Cioni & Strambini, 2002).
Evidently the cell implements special adaptive mechanisms promoting the functional reorganization of vital systems in 2Н2О. Thus, for the normal synthesis and function in D2О of such vital compounds as nucleic acids and proteins contributes to the maintenance of their structure by forming hydrogen (deuterium) bonds in the molecules. The bonds formed by deuterium atoms are differed in strength and energy from similar bonds formed by hydrogen. Somewhat greater strength of 2Н–O bond compared to 1H–O bond causes the differences in the kinetics of reactions in H2O and 2Н2О. Thus, according to the theory of a chemical bond the breaking up of сovalent 1H–C bonds can occur faster than C–2Н bonds, the mobility of 2Н3O+ ion is lower on 28.5 % than Н3O+ ion, and О2Н- ion is lower on 39.8 % than OH- ion, the constant of ionization of 2Н2О is less than that of H2O (Mosin et al., 1999b). These chemical-physical factors lead to slowing down in the rates of enzymatic reactions in D2О (Cleland, 1976). However, there are also such reactions which rates in 2Н2О are higher than in H2O. In general these reactions are catalyzed by 2Н3O+ or H3O+ ions or O2Н- and OH- ions. The substitution of 1H with 2Н affects the stability and geometry of hydrogen bonds in an apparently rather complex way and may, through the changes in the hydrogen bond zero-point vibration energies, alter the conformational dynamics of hydrogen (deuterium)-bonded structures of DNA and proteins in 2Н2О. It may cause disturbances in the DNA-synthesis during mitosis, leading to permanent changes on DNA structure and consequently on cell genotype (Lamprecht et al., 1989). Isotopic effects of deuterium, which would occur in macromolecules of even a small difference between hydrogen and deuterium, would certainly have the effect upon the structure. The sensitivity of enzyme function to the structure and the sensitivity of nucleic acid function (genetic and mitotic) would lead to a noticeable effect on the metabolic pathways and reproductive behaviour of an organism in the presence of 2Н2О (Török et al., 2010). And next, the changes in dissociation constants of DNA and protein ionizable groups when transferring the macromolecule from H2O into 2Н2О may perturb the charge state of the DNA and protein molecules. All this can cause variations in nucleic acid synthesis, which can lead to structural changes and functional differences in the cell and its organelles. Hence, the structural and dynamic properties of the cell membrane, which depends on qualitative and quantitative composition of membrane’s fatty acids, can also be modified in the presence of 2Н2О. The cellular membrane is one of the most important organelles in the bacteria for metabolic regulation, combining apparatus of biosynthesis of polysaccharides, transformation of energy, supplying cells with nutrients and involvement in the biosynthesis of proteins, nucleic acids and fatty acids. Obviously, the cell membrane plays an important role in the adaptation to 2Н2О. But it has been not clearly known what occurs with the membranes - how they react to the replacement of protium to deuterium and how it concerns the survival of cells in 2Н2О-media devoid of protons.
7. Conclusion
The successful adaptation of organisms to high concentration of 2H2O will open a new avenues of investigation with using [U- 2H]labeled macromolecules could be isolated from these organisms. For example, fully deuterated essential macromolecules as proteins and nucleic acids will give promise of important biological, medical and diagnostical uses. Modern physical methods of study the structure of [U- 2H]labeled macromolecules, particularly three-dimentional NMR in a combination with crystallography methods, X-ray diffraction, IR-, and CD- spectroscopy should cast new light on many obscure problems concerning with the biological introduction of deuterium into molecules of DNA and proteins as well as the structure and the function of macromolecules in the presence of 2H2O. The variety of these and other aspects of biophysical properties of fully deuterated macromolecules in the presence of 2H2O remain an interesting task for the future.
References
Cioni, P. & Strambini, G.B. (2002) Effect of heavy water on protein flexibility. Biophysical J., 82(6): 3246–3253.
Cleland W.N. (1976) Isotope effects on enzyme-catalyzed reactions. W.N. Cleland, M.N. O'Leary & D.D. Northrop (eds.). Baltimore, London, Tokyo, University Park Press, 303 p.
Crespi H.L. (1989) Fully deuterated microorganisms: tools in magnetic resonance and neutron scattering. Synthesis and applications of isotopically labeled compounds. in: Proceedings of an International Symposium. T. Baillie & J.R. Jones (eds.) Amsterdam: Elsevier, pp. 329–332.
Den’ko, E.I. (1970) Influence of heavy water (D2O) on animal, plant and microorganism’s cells. Usp. Sovrem. Biol., 70(4): 41–49.
Eryomin, V.A., Chekulayeva, L.N. & Kharatyan, F.F. (1978) Growth of Micrococcus lysodeikticus on a deuterated medium. Microbiologia, 14: 629–636 [in Russian].
Ignatov I. & Mosin, O.V. (2013a) Possible processes for origin of life and living matter with modeling of physiological processes of bacterium Bacillus subtilis in heavy water as model system. Journal of Natural Sciences Research, 3(9): 65-76.
Ignatov, I. & Mosin, O.V. (2013b) Modeling of possible processes for origin of life and living matter in hot mineral and seawater with deuterium. Journal of Environment and Earth Science, 3(14): 103-118.
Ignatov, I. & Mosin, O.V. (2013c) Structure of water for origin of life and living matter. Naukovedenie, 2: 1-16, ISSN 2223-5167, online (March-April 2013): naukovedenie.ru/PDF/05tvn213.pdf.
Ignatov, I. & Mosin, O.V. (2013d) Structural mathematical models describing water clusters. Journal of Mathematical Theory and Modeling, 3(11): 72-87.
Katz, J.J. (1960) The biology of heavy water. Scientific American, pp. 106-115.
Kushner, D.J., Baker, A. & Dunstall, T.G.. (1999) Pharmacological uses and perspectives of heavy water and deuterated compounds. Can. J. Physiol. Pharmacol., 77(2): 79–88.
Laeng, R.H., Mini, R.L., Laissue, J.A. & Schindler R. (1991) Radioprotection of cultured cells by preincubation in medium containing deuterium oxide. Int. J. Radiat. Biol., 59(1): 165–173.
Lamprecht, I. Schroeter, D. & Paweletz, N. (1989) Disorganization of mitosis in HeLa cells by deuterium oxide, European journal of cell biology, 50(2): 360-369.
LeMaster, D.M. (1990) Uniform and selective deuteration in two-dimensional NMR studies of proteins. Ann. Rev. Biophys. Chem., 19: 243–266.
Lis, G., Wassenaar, L.I. & Hendry, M.J. (2008) High-precision laser spectroscopy D/H and 18O/16O Measurements of microliter natural water samples. Anal. Chem., 80(1): 287–293.
Lobishev, V.N. & Kalinichenko, L.P. (1978) Isotopic effects of D2O in biological systems. Moscow, Nauka, 215 p.
MacCarthy, P. (1985) Infrared spectroscopy of deuterated compounds: an undergraduate experiment. J. Chem. Educ., 62(7): 633–638.
Michel, F., Altermatt, H.J., Gebbers J.O. et al. (1988) Radioprotection by pretreatment with deuterated water: cytokinetic changes in the small intestine of the mouse. Virchows. Arch. B. Cell. Pathol. Incl. Mol. Pathol., 54(4): 214–220.
Mosin, O.V. (1996a) Studying of methods of biotechnological preparation of proteins, amino acids and nucleosides, labeled with stable isotopes 2H, 13C and 15N with high levels of isotopic enrichment: autoref. disser. thesis PhD: Moscow, M.V. Lomonosov State Academy of Fine Chemical Technology, 26 p.
Mosin, O.V., Skladnev, D.A., Egorova, T.A. & Shvets, V.I. (1996b) Mass-spectrometric determination of levels of enrichment of 2Н and 13С in molecules of amino acids of various bacterial objects. Bioorganic Chemistry, 22(10–11): 856–869.
Mosin, O.V., Skladnev, D.A. & Shvets, V.I. (1996c) Methods for preparation of proteins and amino acids, labeled with stable isotopes 2Н, 13С and 15N. Biotechnologia, 3: 12–32 [in Russian].
Mosin, O.V., Skladnev, D.A. & Shvets, V.I. (1998) Biosynthesis of 2H-labeled phenylalanine by a new methylotrophic mutant Brevibacterium methylicum. Bioscience, biotechnology, and biochemistry, 62(2): 225–229.
Mosin, O.V., Skladnev, D.A. & Shvets, V.I. (1999a) Incorporation of [2,3,4,5,6-2H]phenylalanine, [3,5-2H]tyrosine, and [2,4,5,6,7-2H]tryptophan into bacteriorhodopsin molecule of bacterium halobium. Applied Biochemistry and Microbiology, 35(1): 34–42.
Mosin, O.V., Skladnev, D.A. & Shvets, V.I. (1999b) Studying physiological adaptation of microorganisms to heavy water. Biotechnologya, 8: 16–23.
Mosin, O.V., Shvets, V.I., Skladnev, D.A. & Ignatov, I. (2012a) Studying of microbic synthesis of deuterium labeled L-phenylalanine by methylotrophic bacterium Brevibacterium Methylicum on media with different content of heavy water. Russian Journal of Biopharmaceuticals, 4(1): 11–22.
Mosin, O.V. & Ignatov, I. (2012b) Isotope effects of deuterium in bacterial and microalgae cells at growth on heavy water (D2O). Voda: Himia i Ecologija, 3, 83–94 [in Russian].
Mosin, O.V. & Ignatov, I. (2012c) Studying of isotopic effects of heavy water in biological systems on example of prokaryotic and eukaryotic cells. Biomedicine, Moscow, 1(1–3): 31–50 [in Russian].
Mosin, O.V., Ignatov, I., Skladnev, D.A. & Shvets, V.I. (2013a) A strain of Gram-positive chemoheterotrophic bacterium Basillus subtilis – producer of [2H]riboxine. Drug development & registration, 4(5): 110–119 [in Russian].
Mosin, O.V., Shvets, V.I., Skladnev, D.A. & Ignatov, I. (2013b) Microbial synthesis of 2H-labelled L-phenylalanine with different levels of isotopic enrichment by a facultative methylotrophic bacterium Brevibacterium methylicum with RuMP assimilation of Carbon. Biochemistry (Moscow) Supplement Series B: Biomedical Chemistry, 7(3): 249–260.
Mosin, O.V. & Ignatov, I. (2013c) Studying the biosynthesis of 2H-labeled inosine by a Gram-positive chemoheterotrofic bacterium Bacillus subtilis B-3157 on heavy water (2H2O) medium. Chemical and Process Engineering Research, 15: 32–45.
Mosin, O.V., Shvez, V.I, Skladnev, D.A., & Ignatov, I. (2013d) Microbiological synthesis of [2H]inosine with high degree of isotopic enrichment by Gram-positive chemoheterotrophic bacterium Bacillus subtilis. Applied Biochem. Microbiol., 49(3): 255–266.
Mosin, O.V. & Ignatov, I. (2013) Microbiological synthesis of 2H-labeled phenylalanine, alanine, valine, and leucine/isoleucine with different degrees of deuterium enrichment by the Gram-positive facultative methylotrophic bacterium Вrevibacterium methylicum. International Journal of Biomedicine, 3(2), 132–138.
Mosin, O.V., Ignatov, I., Skladnev, D.A. & Shvets, V.I. (2014) A strain of Gram-positive facultative methylotrophic bacterium Brevibacterium methylicum – producer of [2H]phenylalanine. Drug development & registration, 1(6): 58-67 [in Russian].
Somlyai, G. (2001) The biological effect of deuterium-depleted water. A possible new tool in cancer therapy. Anticancer Research Intern. J., 21(3): 23–33.
Török, G., Csík, M., Pintér, A. et al. (2000) Effects of different deuterium concentrations of the media on the bacterial growth and mutagenesis. Egészségtudomány/Health Science, 44: 331-338.