Origin of Life and Living Matter in Hot Mineral Water
Prof. Dr. Ignat Ignatov
Ass. Prof. Oleg Mosin PhD
Additional information about the report:
www.medicalbiophysics.dir.bg/en/water_memory.html
1. Dissipative structures of Prigozhin. Origination of life and living matter in hot mineral water (Ignatov, Mosin, 2009)
New achievements in the studies of water structure allow to better analyze the conditions for the emergence of life. It is difficult to presume that life has originated in chaotic
water (Ignatov, Mosin, 2010). Living organisms and water (Antonov, Galabova, 1992) are complex self-organizing systems. Schrödinger clearly defines that living organisms reduce their own entropy at the expense of increasing the entropy of the environment.
Self-organization can most logically explain the origin of living matter. The term itself was introduced in 1947 by Ashby. The dissipative structures of Prigozhin and the hyper cycles of Eigen show that life is most probably not the result of chaotic processes. Prigozhin demonstrated that the formation of dissipative structures and the increase of their complexity is associated with changes in entropy.
For their achievements, both scientists separately received the Nobel Prize.
In late 2009 and early 2010 Ignatov and Mosin carried out experiments with control deionized
water, mineral water, sea water, and mountain water from Bulgaria. Water from karst springs was also studied. The experiments were made with Antonov’s device for spectral analysis of water. Cactus juice was studied too (Ignatov, Mosin, 2009). The cactus was selected as a model system because the plant contains about 90% water. Also, photosynthesis is carried out by the enlarged stems, which serve for storage of water as well. Mineral water from different springs was examined.
Closest to the spectrum of sap, however, is the spectrum of mineral water, which reacts with calcium carbonate (СаСО3). Karst springs have a similar spectrum. Closest to the spectrum of sap is the spectrum of karst springs water, which interacts with mineral water.
In nature there are places with different water and identical external conditions. 25 km away from Teteven there is the Zlatna Panega karst spring with a temperature from 10 to 12,5 °C. Three mineral springs with an average temperature of 21 °C pour into the lake formed by the spring. Only a few kilometers away is the Vit river with an average temperature of around 15 °C. Photographs demonstrate the obvious difference between the plant life in the water of the river and the karst spring. This is visible proof of the optimally good place for the active life of algae under identical external conditions. The difference is only in the structure of water.

Karst and mineral spring, vegetation Zlatna Panega, Teteven district, Bulgaria
Photo: Alexander Ignatov

Vit river, Teteven district, vegetation 7 km away from Zlatna Panega, Bulgaria
Photo: Alexander Ignatov
Close in value peaks in the amplitudes of the spectra of sap and of karst and mineral springs, which interact with calcium carbonate are observed. Peaks are found at -0.1112, -0.1187, -0.1262, -0.1287 и -0.1387 еV. The average energy of the hydrogen bonds between water molecules in the creation of cluster formations is measured. Similar amplitudes in the spectrum between the sap and the mountain and sea water is observed at -0.1362 еV. The spectrum of the control deionized
water is substantially different from that of sap, mineral and mountain water.
There are three components that are exceptionally important before we proceed further. Modern physics can determine the composition of mineral water to the nearest atom. If, however, we mix these ingredients in deionized water, we will not obtain mineral water. Which are the factors that preserve information in a self-organizing
structure. The more logical outcome is for it to self-destruct. Naturally all these processes have occurred due to energy from the environment.
Oparin, theoretically, and Miller, experimentally, have indicated the possibility for formation of organic molecules in the conditions of the young Earth.
Experiments and analyses indicate close quantum-mechanical distribution according to energies of water molecules between a liquid, which is directly connected with the plant, and mineral and karst water. Mineral water in springs and geysers flows with a nearly constant composition, and long enough. In contact with calcium carbonate (CaCO3), sulphur (S) and other elements (Na, K, Ca, Mg, etc.), stable cluster formations are established. And since the external factor is repeated, they are more difficult to destroy. Energy required for maintaining the self-organizing structures directly depends on the heat from the volcanic activity and the magma. It also depends on the solar energy. Entropy depends on the amount of heat and the self-organized structure does not self-destruct because the energy balance is maintained.
2. The stromatolites and protocell of Sugawara - evidence for the origination of life in hot mineral water. Possible processes for the origination of life.
A proof of this is that the oldest traces of photosynthesizing organisms are the stromatolites. The oldest stromatolites were found in Greenland. They are 3.5 billion years old. They have a complex laminar structure of calcium carbonate and extract hydrogen from water. Even today they exist in Shark-Bay, Australia. Perhaps there has been mineral water at their very inception at the bottom of the water basins, or after their creation close to the shore, they have spread in water basins. Life has been discovered at the bottom of the oceans under extremely harsh conditions and it was near hot mineral springs.

Dwarf Planet 2007 OR10
One of the unique places in the Solar system is the satellite of Saturn – Enceladus. There the magma maintains liquid water and there is a probability for the existence of life. One of Jupiter’s moons – Europa – holds similar expectations. In August 2011, red-colored ice was discovered on the dwarf planet 2007 OR10 in the Solar system. Analyses showed that these are carbon polymers. The explanation is a beginning of origination of life, when there have been active volcanoes on the planet as an energy source. Solar activity here is too low to sustain life activity.
The Nature
journal indicates that there is new evidence for the origination of vegetation 1.5 billion years ago. You can obtain an impression of the first species of vegetation at the Teteven waterfall.
The plant cell cannot exist without the following organogenic elements C, H, N, P, O, S and the elements Na, K, Ca, Mg, Cl, B, etc.
Let’s review the following reactions:
(1) CO2 + 4H2S + O2 = CH2O + 4S + 3H2O
(2) СаСО3+ HOH + СО2 = Ca(HCО3)2
The first equation shows how some chemosynthetic bacteria use energy from the oxidation of hydrogen sulfide (H2S) to sulfur (S).
The second equation is related to one of the most common processes in nature.
In the presence of water and carbon dioxide, calcium carbonate transforms into calcium hydrogencarbonate.
In the presence of hydroxyl OH- ions, the cellular processes are activated. Kagava demonstrates that an effect of improving the conductivity of the cell membrane is observed. The valid reaction is:
(3) CO2 + ОН- = HCО3-
(4) 2 HCO3- + Ca2+ = CaCO3 + CO2 + H2O
It is assumed that the second reaction has been valid upon the origination of the stromatolites.
Contemporary chlorophyll contains the elements C, H, O, N, Mg.

Karst spring bubbles, Zlatna Panega, Teteven district, Bulgaria
Photo: Alexander Ignatov
When considering the issue of self-organization in nature, there is an exceptionally interesting example found in the karst springs in Zlatna Panega, Teteven district. Algae are surrounded with bubbles 3-5 mm in size. These bubbles are retained long enough – from hours to days. Water itself, which is similar in its spectrum to plants, seeks
to preserve the self-organizing structures. During the study, the ambient temperature was 5 °C. It is known that if Ca2+ ions are added to a solution of pectin molecules, the solution is gelatinised. The reason is that Ca2+ ions bind to pectin molecules and cellulose microfibrils are formed. There is evidence that this kind of Ca2+ - sutures play a crucial role in the unification of the different components of the cell wall and influence its compactness and strength. Cytoplasm consists of 99% water, ions and other elements that form its basic structure.
In 2003, Arge and McKinnon received the Nobel Prize in chemistry for the discovery of water channels for water penetration into the cells. The aquaporin protein that regulates water exchange plays a role here. By increasing the concentration of calcium ions this process is more active.
Cabanero and team carried out experiments, which show the role of Ca2+ in the permeability of the cell wall of plant cells.

Organism with a silicon skeleton
In the most ancient organisms in evolution a silicon skeleton is observed. These are the sea sponges, the radiolaria and the diatomeae. Silicon was gradually displaced by the more active element calcium. The organisms reached the silicon-calcium skeleton and the most evolutionarily advanced of them – the calcium skeleton.
Molecular biology, almost like a dogma, adopts the model for the structure of the cell membrane. The main part of the membrane is made up of a double phospholipid layer.
In the Sargasso Sea a phenomenon is observed. Membranes of blue-green algae cells are made of phosphorus-containing lipids. The reason is that there is almost no phosphorus and nitrogen in the Sargasso Sea. The unicellulars have found a smart way to use sulfur instead of phosphorus in their lipid membranes. Apparently, in self-organization
the structures choose the optimal components for them.
However, let's look at the structuring of water clusters.
3. Stromatolites in the Glacier National Park and Yellowstone National Park in the USA (David Ward). Photocell of Tadashi Sugawara

Yellowstone National Park, USAThe evidence shown indicates that the emergence of life depends on the properties and structure of water and also on additional conditions. Mineral water, which interacts with calcium carbonate is closest to these conditions and has left a trace in plants with its structure, and entropy. Next in line with regard to quality are sea and mountain water (Ignatov, Mosin, 2010). In warm and hot mineral waters the peaks in the differential non-equilibrium energy spectrum (DNES) (Antonov, 1993) are more pronounced in comparison to the peaks in the same water with a lower temperature. This signifies that there is more energy for the preservation of a self-organized structure. The spectral range of DNES is in the middle infrared range from 8 to 14 micrometers. There is the earth atmosphere’s window of transparency for the electromagnetic radiation in the close and middle infrared range. In this interval energy is radiated from the Sun towards the Earth, and from the Earth towards surrounding space. Water changes with a cosmic rhythm. The likelihood of origination of life is biggest in warm and hot water with a specific structure (Ignatov, Mosin, 2009, 2010). In January 2010, American scientist David Ward and colleagues described fossilized stromatolites in the Glacier National Park in the USA. They are studying microbes in Yellowstone National Park in USA, which are building stromatolites in hot water similar like ancient organisms. Rotorua in New Zealand is a similar place. Stromatolites have lived in warm and hot water in zones of volcanic activity. Warm and hot waters can be heated by magma as well. Their age is 3.5 billion years in Greenland. These are the first organisms with fossil evidence. This is a confirmation and of the concept based on biophysical analyses for origination of life in warm and hot mineral waters and geysers (Ignatov, Mosin, 2009, 2010). In June 2010, an article with this evidence was published at an Euromedica congress in Hanover, Germany. In September 2010, the American scientists Stockbridge, Lewis, Yung Yuan and Wolfenden published an article with the popular title Is the Origin of Life in Hot Water?
. They consider the probability of faster biochemical reactions in hot water. We think that in the beginning of evolution there was much more deuterium in water and this is a significant fact regarding its information abilities about the preservation of life.
In September 2011 a team of Japanese scientists led by Tadashi Sugawara also brought us closer to the secret that life has originated in warm or, more likely, hot water. They have created a proto cells, witch are similar of bubbles. For this purpose, they have made an aqueous solution of organic molecules, DNA and synthetic enzymes. The solution was heated to a temperature close to water’s boiling point – 95 degrees. Then its temperature was lowered to 65 degrees. A formation of a proto cells wits membrane was also observed. These proto cells are multiplying. This is a step for creation of synthetic cell. This laboratory experiment is an excellent confirmation of the possibility that life has originated in hot water (Ignatov, 2010), (Ward, 2010). Upon changing the temperature is changing the middle energy of hydrogen bonds among water molecules, as an information bearer of life. (Ignatov, Antonov, 1998).
4. Proofs of Marie-Laure Pons from Greenland. Rupite, Bulgaria

Movement of hot mineral water rich in minerals

In hot mineral water is breathing
air
In 2011 the team from the Laboratory of Geology in Lyon, France with head Marie-Laure Pons were studied some of the oldest rocks in the planet and found the mineral serpentinite. It was previously thought that the first living creatures evolved in geysers. The French scientists were proved that in Greenland the water was rich in carbonates, not too acidic. The temperatures would have ranged from 100 to 300°C. The process in geysers also had been possible in the sea. These new results are an excellent confirmation of the possibility that life has originated in hot mineral water (Ignatov, 2010), (Ward, 2010). Also is very important that this process was connected with carbonates (Ignatov, Antonov, Galabova, Stoyanov, 2001).
In Rupite, Bulgaria is possible observation of scenario
for origination of life in hot mineral water near dead volcano.
In 2012 Brian Switek was published in Nature the report Debate bubbles over the origin of life
. In 2009 Mulkidjanian and Michael Galperin were published the report that the first cells were developed in zinc-rich environments. Mulkidjanian informed: Тhe cytoplasm is rich in potassium, zinc, manganese, and phosphate ions, which are not widespread in marine environments, and has lower amounts of sodium ions than outside.

Disposition of minerals and formation of structures
Irene Klotz shows in Discovery News the opinion of Nobel holder Jack Szostak The accumulation of organic compounds in endorheic (free-standing) ponds is also easier to imagine than in the ocean, and geothermally active areas provide numerous advantages
.
The following experiment was conducted in laboratory settings (Ignatov, 2012). The first living structures originated in the warm and hot mineral water with more bicarbonate ions (HCО3-), metal ions (Na, Ca, Mg, Zn, К etc.) and deuterium molecules (Ignatov, Mosin, 2012). There was a gas discharge (lightning) in the ancient atmosphere and close to the water. The water in the electrode of the Kirlian device has a similar composition. It has electric discharge, like in the ancient atmosphere. Biophysical fields from a biological object and gas discharge are also emitted in a specific zone in the center of the electrode. A structure sized 12/14 mm was formed as a result of the experiment. It resembles a small jelly-fish. No structure is organized in a control sample with water from an electrode. Before being placed in the electrode, water is heated to boiling point and then cooled down. With the increase of the gas discharge duration, the structure is also increased. This experiment demonstrates that under specific external conditions, the organization of structures in the water becomes reality. Water was heated by the magma.
5. Results with Infrared spectroscopy for the spectrum of water for origin of life and living matter (Ignatov, Mosin, 2009)
Furthermore, we have carried out the research of mineral, sea and mountain water from Bulgaria by IR-spectroscopy method of differential non-equilibrium energy spectrum (DNES) relative to the control – deionized water (fig. 2, curves 1-5, the table). In experiments were investigated samples of water from karst springs. Also IR-spectra of cactus juice were investigated by DNES method (fig., curve 1). The cactus was selected as a model system because the plant contains about 90% water. The closest to the IR-spectrum of cactus juice was the IR-spectrum of the mineral water contacting with СаСО3 (fig., curve 2). IR-spectra of plant juice, mineral water and water of the karst springs have magnitudes of peaks in IR-spectra at -0,1112; -0,1187; -0,1262; -0,1287 and -0,1387 eV, accordingly. Similar peaks in the IR-spectrum between cactus juice, mountain and sea water were detected at -0,1362 eV. The IR-spectrum of the control sample of deionized water (fig., curve 5) was substantially different from the IR-spectrum of sea mineral and mountain water. The values of average energy (∆EH... O) of hydrogen Н…O-bonds between molecules H2O in the process of formation of (H2O)n associates, measured by the DNES method were measured at 0,1067±0,0011 eV.
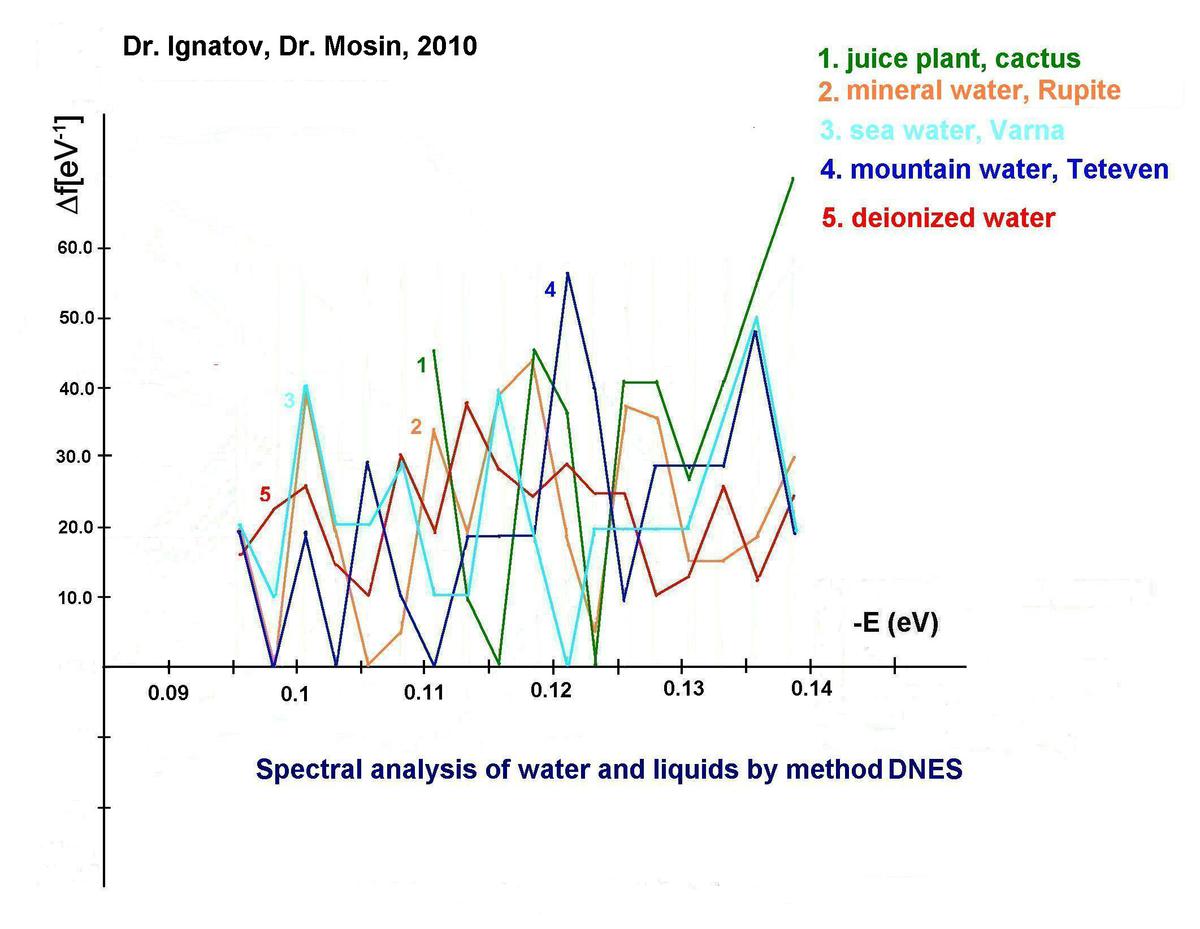
DNES spectra of water of various origin: 1 – cactus juice; 2 – mineral water Rupite (Bulgaria); 3 – sea water (Varna, Bulgaria); 4 – mountain water (Teteven, Bulgaria); 5 – deionized water (control).
The table. Characteristics of IR-spectra of water obtained by DNES-method.
- -Ex(eV) Cactus juice; -E (eV) Mineral water Rupite; -E (eV) Sea water; µm; cm-1
- 0,1112; 0,1112; ; 11,15; 897
- 0,1187; 0,1187; ; 10,45; 957
- 0,1262; 0,1262; ; 9,83; 1017
- 0,1287; 0,1287; ; 9,64; 1037
- 0.1362; ; 0,1362; 9,10; 1099
- 0,1387; 0,1387; ; 8,95; 1117
The function of the distribution of energies ∆f was measured in reciprocal electron volts
(eV-1). It is shown of which spectrum values -E in eV was observed the biggest local maximums of this function.
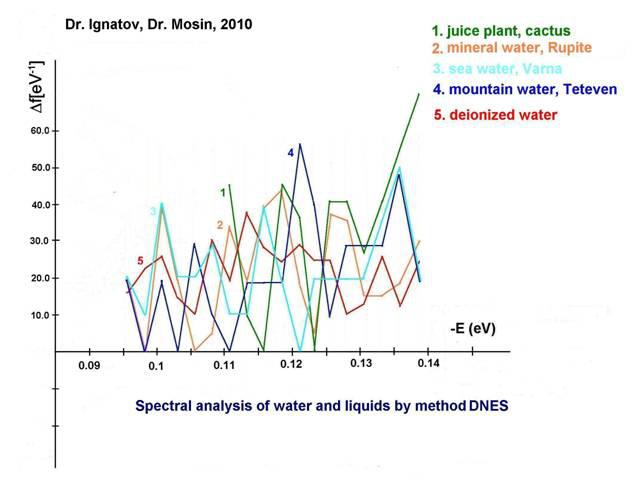
Results of infrared spectrum of Rupite, Bulgaria
The spectrum of water in Rupite has been studied with infrared spectroscopy method with the device Thermo Nicolet Avatar 360 Fourier-transform IR. The study was carried out by Christina Chakarova, Bulgarian Academy of Sciences. At 9.7 μm or -0.1287 eV obtained value of local maximum. A value of local maximum at 9.64 μm or -0.1278 eV was obtained with the infrared spectroscopy method DNES. The statistical reliability of the DNES method is ± 0.0011 eV. The local maximum obtained with the DNES method at 9.83 μm (-0.1262 eV) and 8.95 μm (-0.1387 eV) are located on the spectral curve of the local maximum 9.7 μm (-0.1287 eV). The data obtained proved that hot mineral alkaline water is preferable for of life. These data also can predict a possible way of transition from synthesis of small organic molecules due to the energy of UV solar radiation and thermal activity to more complex organic molecules as protein and nucleic acids. The important factor in reaction of condensation of two molecules of amino acids is allocation of H2O molecule when peptide chain is formed. As reaction of polycondensation of amino acids is accompanied by dehydratation, the H2O removal from reactional mixture speeds up the reaction rates. This testifies that formation of organic forms may occur nearby active volcanoes, because at early periods of geological history volcanic activity occurred more actively than during subsequent geological times. However, dehydratation accompanies not only amino acid polymerization, but also association of other blocks into larger organic molecules, and also polymerization of nucleotides into nucleic acids. Such association is connected with the reaction of condensation, at which from one block removes proton Н+, and from another – hydroxyl group (OH-) with formation of H2O molecule.
The possibility of existence of condensation-dehydratation reactions under conditions of primary hydrosphere was proven by Calvin in 1965. From most chemical substances hydrocyanic acid (HCN) and its derivatives – cyanamid (HNCN2) and dicyanamid (HN(CN)2) possess dehydratation ability and the ability to catalyze the process of linkage of H2O from primary hydrosphere [12]. The presence of HCN in primary hydrosphere was proven by Miller's early experiments. Chemical reactions with HCN and its derivatives are complex with chemical point of view; in the presence of HCN, HNCN2 and HN(CN)2 the condensation of separate blocks of amino acids accompanied by dehydratation, can proceed at normal temperatures in strongly diluted H2O-solutions. Furthermore, polycondensation of amino acids in the presence of HCN and its derivatives depends on acidity of water solutions in which they proceed. In acid water solutions (рН 4-6) these reactions do not occur, whereas alkaline conditions (рН 8–9) promote their course. There has not been unequivocal opinion, whether primary water was alkaline, but it is probable, that such a value of рН possessed mineral waters adjoining with basalt, and these reactions could occur at contact of water with basalt rocks.
6. Atmosphere on the young Earth. Water on the young Earth with more deuterium molecules (Mosin, 1996)
Let us go millions or even 3-4 billion years back. The air of the young Earth was different from the present. It contained little oxygen. It consisted mainly of hydrogen, water vapor, ammonia (NH3), methane (CH4) and others. Also, the strong volcanic activity has emitted into the atmosphere nitrogen (N), hydrogen sulfide (H2S) and sulfur dioxide (SO2). Even the water was different. In 1996 Mosin made the assumption that water contained much more deuterium in the early evolution. According to this review of the Russian scientist, life began to emerge in water with more water molecules containing deuterium. This water is known as heavy. Heavy water was discovered by American scientist Urey in 1939. The water molecule with two atoms of deuterium isotope is heavier and more electronegative. This water is also colorless. In it, however, chemical reactions proceed more slowly. Only the simplest organisms can exist in 75% water with deuterium. Research of American scientists with ultraviolet telescope showed that the Universe has more deuterium than what we have assumed thus far. This evidence changes a series of views on the evolution of stars and galaxies according to American scholar Lynskey. Interestingly, however, hydrogen bonds in water with deuterium in the water molecules are more stable. This means that in the primary water on Earth the self-organizing structures have managed to preserve themselves for a longer time. There is evidence that during this period there has been a process of structuring of organic molecules.
Meteorites and comets give us much information about the origination of the Solar system. Most impressive is the Murchison meteorite that fell in Australia in 1962. 18 amino acids, which are not of terrestrial origin, have been found in its analysis.
The amino acid glycine, which is the basis of protein, has been found in the tail of the Wild-2 comet in 2009. These scientific advances show that the building blocks
of life have existed in the origination of the Solar system and the Earth. On our planet the molecules fell in the aquatic environment of evolution. Amino acids and water clusters began to self-organize. This activity was maintained with energy from the magma, the volcanic activity and the solar activity.
The shown analyzes of the water spectrum indicate that most favorable for the development of life are the mineral waters that interact with calcium carbonate and then - sea water (Ignatov, Mosin, 2010). The spectrum of karst water is also very close to that of plants. The oldest evidence of living organisms with complex laminar structure of calcium carbonate date 3.5 billion years back. These organisms – stromatolites – soak up silicon dioxide. Karst water itself contains calcium carbonate, actively interacts with living matter and carries information about life from a later geological period. The mixture of karst and mineral water from Zlatna Panega, Bulgaria which shows us the spectrum of mineral water, which interacts with calcium carbonate from a karst spring, is unique. Over 30 springs of mineral water containing calcium and carbonate ions have been researched and have a spectrum that is close to that of the water from Zlatna Panega.
The photograph shows the richness of Chlorella algae in the Rupite, Bulgaria. The place is located near the bottom of a dead volcano. At a temperature of 75 °C the flora boils with life. The water in Rupite is characterized with a greater amount of bicarbonate ions (HCO3-) (1320 -1488 mg / l, Vladeva). It is assumed for these ions, like for calcium ions (Ca2+) that they have been involved in the formation of stromatolites (4). Apparently, the larger amount of carbon atoms is essential for the self-organization of a living cell of molecules. This water contains both sodium, calcium and magnesium ions as well as hydrogen sulfide (H2S), metasilicic acid (H2SiO3), carbon dioxide. Water in Rupite approaches in composition the water in which life has originated. Only a few kilometers away is the river Struma and no such diversity of algae is observed there. Time will tell whether in Rupite there have been stromatolites. It is possible that natural disasters have destroyed them. In Bulgaria stromatolites have been found in the Pobitite Kamani location near Beloslav, Varna district (Nachev). The place is near the Black Sea coast. There are many mineral springs in this area.

Algae, mineral water, 75 °C, Rupite, Vanga’s place
photo: Alexander Ignatov
In the beginning, on the Earth there was a more intense volcanic activity, even at the bottom of the primary ocean. Then silicon was in greater quantity and it interacted with water. Therefore, it was absorbed into living organisms. At a later stage, the organisms began to absorb calcium from the limestone rocks. Organisms with silicon skeleton reach a size of up to several tens of micrometers.
Life on Earth has started to select
silicon and calcium to build the cell membrane. As shown by the membrane composition of sulfur in the Sargasso Sea, living matter seeks to preserve itself on the basis of self-organization. This is dependent on external conditions and recurring factors as is the water with a specific structure (Dr. Ignatov, 2010).
In 2010 the „Science” journal published information about a scientific experiment of U.S. NASA scientists who succeeded in “making” bacteria replaces phosphorus with arsenic at the Mono Lake in the USA. The bacterium has managed to replace the phosphorus even in the DNA. Generally phosphorus is considered one of the 6 irreplaceable elements of life. This discovery shows how flexible life is.
The first photograph shows one of the stones with minerals that have been found by Tsolo Petkov in Stara Planina mountain near town of Shivachevo. The second photograph shows the stones with minerals that have been found by Ignatov at the Bulgarian Black Sea coast. The minerals contain silicon (Si), magnesium (Mg), calcium (Ca), etc. They were tested in the Eurotest Control Laboratory, Sofia, Bulgaria.


Photography with minerals,
Dipl. Eng. Petkov©, Dr. Ignatov
In the early stages of the Earth's development, silicate minerals were able to participate in the origination of life on Earth. The first biological molecules such as ribose, which is part of the DNA composition, could also form at normal temperature. In this process the silicate ions are catalysts and stabilizers of the process. This reaction is called formose and has been studied mainly by Joseph Lambert. This process is possible on the surface of carbonate-clay cliffs. The medium of the formose reaction is alkaline and needs the presence of sodium ions. Mineral water, which interacts with calcium carbonate and contains sodium and others ions, is alkaline and is the information medium for the structuring of a living cell.
An interesting phenomenon is observed in The Black Sea. Below 200 m depth there is hydrogen sulphide (H2S) in the water. In the early evolution some chemosinthetic bacteria use energy from the oxidation of hydrogen sulphide (H2S) to sulphur (S). Perhaps the Black Sea has preserved a memory of this primary ocean.
Gradually in nature there began a cleansing
of the deuterium from the water. According to Mosin, there are two very important processes. The Earth's atmosphere had no protective ozone layer and was hotter than the present one. Then the volcanic geothermal and electrical processes in the atmosphere saturated it with heavy water vapors (Mosin, 1996). According to astronomers, the Earth's gravitational field is not strong enough to hold the lighter hydrogen and it is dissociated in interplanetary space. This occurs faster than with the heavier deuterium, which is absorbed in the water. At a later stage, water started cleansing the deuterium through natural rocks.
Upon reduction of deuterium in water, evolution is faster. Had the process of cleansing the deuterium not started on the Earth, evolution could have remained frozen
at a very low stage.
7. Cells in heavy water
In 1978 Eryomina and Chekulaev published data from electronic microscope of deuterium cells of the bacterium M. Lysodeikticus.
The deuterium cell is larger and a modification of the direction of division. The cell membrane also changes, and it reacts first to the impact of heavy water.
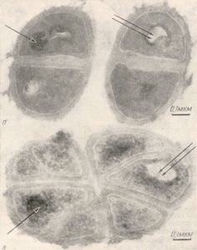
In 1972, Crespi and Katz demonstrated that the increase of the percentage content of deuterium in water reduces plant growth. In 1996 Mosin proved that the alteration of metabolisms in the process of adaptation to heavy water leads to cellular death. With over 50% of deuterium in the water molecules, plants die. Experiments conducted by Mosin show that microorganisms can live in 98% heavy water. Regarding the storage of information from clusters of water molecules, it is considered that a major role in the origin and self-organization of living cells belongs to the deuterium bonds (Mosin, 1996, Ignatov, 2010).
In an experimental plan tap water contains deuterium of more than 100 g. per tonne. Mountain water contains small amounts of deuterium. These two types of water have not existed at the origin of life. Representatives of the 
To the left is the plant that is watered with tap water.
To the right is the plant, which is watered with mountain water from Teteven, Bulgaria
(Dr. Ignatov, Tsvetkova 2010)Club of Medical Biophysics from Teteven, Bulgaria under the direction of Dr. Ignatov held the following scientific experiment. They watered identical seeds with tap water and mountain water. Plants watered with mountain water grew faster. The larger amount of deuterium reduces the permeability of the cell membrane. The figure shows the result with maize after 1 month of watering (Ignatov, Tsvetkova 2010).
Scientifically reliable results were demonstrated by Klima in the growth of plants with different water. When we do research on water, we face its unique properties, which are related to the origination of life.
8. Experiments of Fox and Miller. Wächtershäuser’s hypothesis
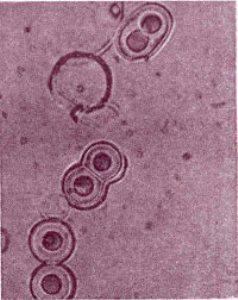
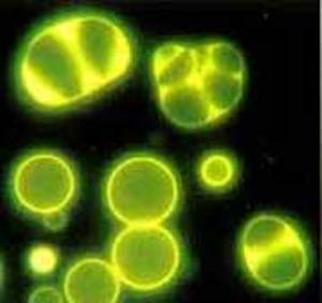
Artificial protein-like compounds, proteinoids that divide themselves with the increase of the alkalinity
of the medium, Fox
In the early 60's of the last century the American scientist Fox did an experiment in which an anhydrous mixture of amino acids is heated to 170 °C. 18 of 23 amino acids found in modern organisms were formed. When Fox washed the hot mixture with water or solutions, structures with a diameter of several nanometers were formed. These are artificial protein-like compounds called proteinoids. Proteinoids have the characteristics of proteins. They have a two-layer shell and upon a change of osmotic pressure they tend to divide themselves. Fox has had a success to combine amino acids into short irregular chains – non-matrix synthesis of polypeptides. He described self-organizing structures similar to coacervates and called them microspheres. The proteinoid microspheres divide themselves with the increase of the alkalinity of the medium.
The California Review indicates that an experiment has been made with water, which is similar in composition to the thermal springs in the primary ocean. Water that is rich in carbon dioxide and iron is heated in a container at 130°C. Chromium and nickel sulphide were added. Small membranes around the molecules have been observed. This experiment demonstrates the process of formation of self-organizing membrane structures.
The experiment was based on the hypothesis of Wächtershäuser for the emergence of living matter in thermal springs. The composition of thermal water is missing calcium and silicon, which are necessary for the skeleton of a living organism. There is a possibility that life forms have originated and have disappeared subsequently. The important thing is that under stable external factors there is a tendency towards self-organization. The authors of the experiment indicate that one of the factors for the structuring of the membrane is the alkaline water. Mineral water, which interacts with calcium carbonate, as well as seawater are alkaline.
The theoretical analysis of Oparin, Miller's experiments, Fox and others clearly show that in nature organic molecules can be structured from inorganic ones. In their experiments the main source of energy is heat. In nature, these are solar radiation and magma energy. Another important conclusion is that the origin of life is possible in an alkaline environment. In all cases self-organization is observed.
9. Self-organization
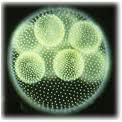
VolvoxVolvox is an example of self-organization of green algae cells. The volvox colony is built from 500 to 20 000 cells. They are associated with cytoplasmic filaments and jelly-like matter. The cells form a globular corpuscle of up to 3 mm.
The development of volvox has amazing features of self-organization, complication, preservation and exchange of information of living matter. In the process of evolution, a development of multicellularity in plants and animals has been repeatedly observed.
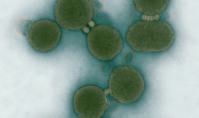
Cell with a genome of another cell, Venter, 2010The ability to transmit information from one cell to another in 2010 was proved with a unique experiment by Craig Venter. The American scientist made a difference in modern science. For the first time in the world a cell is living with the genome (the bio computer) of another cell. Only Nature has created life to the level of water cell, atoms and molecules from the environment.
10. Where in the Solar system is life possible?
The human mind seeks to return to the beginning of life on the basis of evidence and model experiments. Now life exists on the earth, in the air and in water.
When considering the origin of life on the young Earth, the first feature is that it has began in an oxygen-free environment. The energy was from electrical discharges, geothermal and solar radiation. This process is shown in model laboratory experiments. It is possible for a life similar to that on Earth to be born under similar conditions and availability of water on another planet. Moons of Saturn can open the door to life on another body in the Solar system. Enceladus has volcanic activity, and on Titan in 2010 there were unexpected results. On the surface of Mars are observed methane pillars that may be a result from the life activity of microbes.

Is there a life on Titan, the biggest satellite of SaturnTitan is one of the 5 largest satellites in the Solar system together with our Moon and three moons of Jupiter - Ganymede, Io and Callisto. However, it is the only satellite
which has an atmosphere. In 2010 the Cassini space probe made some unique photographs. The presence of life forms that breathe in the atmosphere of Titan is very possible. Perhaps they use the surface chemical compounds, thereby obtaining the necessary energy. NASA scientist Chris McKay stated: "We believe that the hydrogen in the atmosphere of Titan is used by biological forms." He still does not preclude the possibility that this may be a completely new form of biological life, different than the one on Earth. Scientists liken Titan to Earth 
Algae and solar energy, Mountain Vitosha, Bulgaria
Photo: Aleksander Ignatovin its early stages in view of the chemical components of these heavenly bodies. Titan is outside the "inhabitation zone” and the temperature is -170 °C. At this temperature water is in the form of ice and methane is liquid. At the moon of Saturn Enceladus there is 91% of water in atmosphere. Under the ice surface there is liquid water. Enceladus is real candidate for extraterrestrial life.
The image creates the feeling how living matter "aims" to mimic the fluid of water as an aspiration towards the foundation of earthly life. The impact of the Sun creates a feeling of energy. Why shouldn’t life on another planet look like this?
Bibliography:
- Fox S.W., Krampitz G. (1964) Catalytic decomposition of glucose in aqueous solution by thermal proteinoids, Nature, Vol. 203, pp. 1362–1364.
- Fox C.W., Wang C.T. (1968) Melanocytestimulating hormone: Activity in thermal polymers of alpha-ammo acids, Science, Vol. 160, pp. 547–548.
- Sugawara T. et al. (2011) Self-reproduction of supramolecular giant vesicles combined with the amplification of encapsulated DNA, Nature Chemistry, Vol. 1127, pp. P. 775–780.
- Ward D. (2010) First Fossil-Makers in Hot Water, Astrobiology magazine, №1.
- Pons M.L. (2011) Early Archean serpentine mud volcanoes at Isua, Greenland, as a niche for early life, Proc. US National Acad. Sciences, Vol. 108, pp. 17639–17643.
- Mulkidjanian, A. Y.; Galperin, M. Y. (2009) 1. On the origin of life in the Zinc world. 2. Validation of the hypothesis on the photosynthesizing zinc sulfide edifices as cradles of life on Earth, Biology Direct.
- Ignatov, I. (2010) Which Water is Optimal for the Origin (Generation) of Life? Euromedica, Hanover, pp 34-37.
- Ignatov, I. (2011) Entropy and Time in Living Organisms, ArchivEuromedica, Hanover, 1st&2nd Edition, pp. 74-75.
- Informationability of Water, Kirlian (Electric Images) of Different Types of Water, Euromedica, Hanover, pp. 62-65.
- Ignatov, I. (2012) Origin of Life and Living Matter in Hot Mineral Water, Conference on the Physics, Chemistry and Biology of Water, Vermont Photonics, USA.
- Ignatov, I., Mosin, O. V (2012) Isotopic Composition of Water and its Temperature in Modeling of Primordial Hydrosphere Experiments, Euro-Eco, Hanover, p. 62.
- Ignatov, I., Mosin, O. V (2012) Isotopic Composition of Water and its Temperature in Modeling of Primordial Hydrosphere Experiments, VIII Int. Conference Perspectives of the Development of Science and Technique, Biochemistry and Biophysics, Vol. 15, pp. 41-49.
- Ignatov, I., Mosin, O. V (2013) Isotopic Composition of Water and its Temperature in Modeling of Primordial Hydrosphere Experiments, Science Review, №1, pp. 17-27.
- Игнатов, И., Мосин, О. В. (2013) Изотопный состав воды и ее температура в процессе эволюционного происхождения жизни и живой материи, Науковедение, Т. 14, №1, С. 1-16.
- Мосин, О. В., Игнатов, И. Изотопные эффекты дейтерия в клетках бактерий и микроводорослей при росте на тяжелой воде (D2O) //Вода: химия и экология. 2012 № 3. С.
- Мосин, О. В., Игнатов, И. Изучение изотопных эфектов тяжелой воды (D2O) в биологических системах на примере клеток прокариот и эукариот//Биомедицина. 2012. Т.1. №1-3. С. 31-50.
- Мосин, О. В., В.И. Швец, Складнев Д.А., И. Игнатов. Микробный синтез дейтерий-меченного L-фенилаланина факультативной метилотрофной бактерией Brevibacterium Methylicum на средах с различными концентрациями тяжелой воде// Биофармацевтический журнал. 2012. Т.4. №1. С. 11-22.
- Мосин, О. В., Игнатов, И. (2012), Загадки ледяных кристаллов, Сознание и физическая реальность, Т. 17, No. 5, С. 21-31.
- Mosin, O. V, Ignatov, I. (2012) Kirlian Effect in Biomedicine Diagnostics and Research of Properties of Biological Objects and water, Biomedical Radio electronics, Biomedical Technologies and Radio electronics, pp. 13-21.
- Mosin, O. V, Ignatov, I. (2012) Structure of Water, Chemistry, Moscow, No. 11, pp. 24-27.
- Gulyaev, Yu. V., Godik, E. E (1990) Physical Field of Biological Objects, Scientific American, №5, p. 75.
- Gulyaev, Yu. V., Godik, E. E. (1983) Physical Field of Biological Objects, Newspaper of USSR Academy of Science. SU, №8, p. 118.
- Ignatov, I. (2005) Energy Biomedicine, Gea-Libris, Sofia.
- Мосин, О. В., Игнатов, И. (2011) Осознание воды как субстанции жизни, Сознание и физическая реальность, Т. 16, No. 12, С. 9-21.
- Игнатов, И., Мосин, О. В. , Изотопный состав воды и долголетие, Науковедение, Т. 14, №1, С. 2-10.
- Игнатов, И., Мосин, О. В., Нанева, К. (2012) Вода в человеческом теле несет информацию о долголетии, Naturopathie, С. 39-41.
- Мосин, О. В., Игнатов, И. (2011) Структура воды и физическая реальность, Сознание и физическая реальность, Т. 16, No. 9, С. 16-31.
- Мосин, О. В., Игнатов, И. (2012) Адаптация к тяжелой воде. Фенотипическое или генетическое явление? Сознание и физическая реальность, Т. 17, No. 4, С. 25-36.
- Мосин, О. В., Игнатов, И. (2011) Разделение тяжелых изотопов дейтерия (D), трития (Т) и кислорода (18О) в водоочистке, Чистая вода – проблемы и решения, No. 3-4, С. 69-78.
- Мосин, О. В., Игнатов, И. (2011) Современные технологии опреснения морской воды, Энергосбережение, No. 4, С. 14-19.