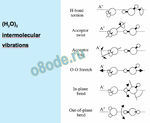
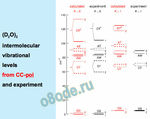
Water
40 years of quantum chemistry from polywater to the real stuff
Почему вода необычная? Исследование маленьких кластеров воды
Ad van der Avoird
Nijmegen, 25 April 2008
Water:
- Most abundant compound on the Earth’s surface
- Principal constituent of all living organisms
- Subject of longer and more vigorous study than any other substance
From: L.C. Allen, The rise and fall of polywater
New Scientist, 16 August 1973.
Anomalies of water
- High boiling point
Large heat of vaporization
Solid (ice) floats on liquid (water)
Ice has many different phases
Liquid density maximum at 4°C
Compressibility decreases with increasing T up to 46.5°C
Large specific heat Cp with minimum at 36°C
Large dielectric constant
High surface tension (highest except metals)
Polywater, also called “anomalous water”
- Freezing point −40◦C
- Boiling point >150◦C
- Density 1.4 g/cm3
- High viscosity
1962 Discovered by N. Fedyakin, Sovjet Union
Then studied by B. Deryagin et al., Moscow
1966 Discussion of the Faraday Society
1966-1972 Intensive studies in UK, USA
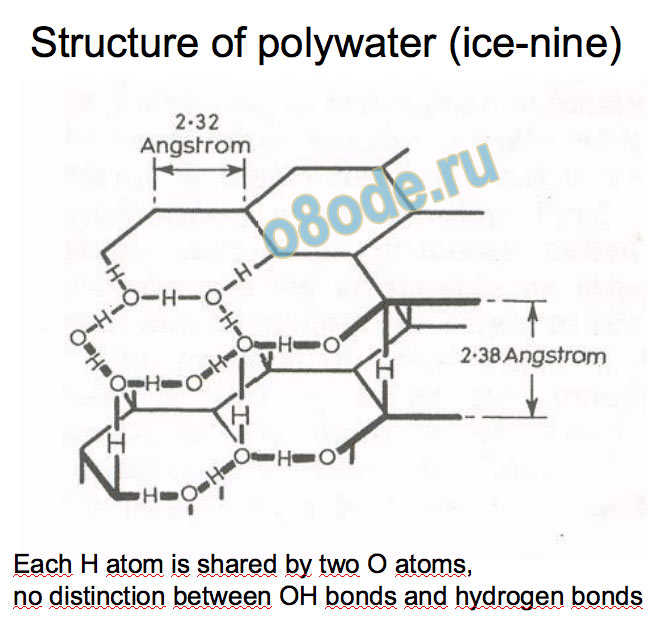
Cat’s cradle (Kurt Vonnegut, 1963):
“There are several ways in which certain liquids can crystallize—can freeze—
several ways in which their atoms can stack and lock in an orderly, rigid way.
Suppose that the sort of ice we skate upon and put into highballs—what we call
ice-l—is only one of several types of ice. Suppose water always froze as ice-l
on Earth because it never had a seed to teach it how to form ice-two, ice-three,
ice-four…? And suppose that there were one form, which we will call ice-nine,
a crystal as hard as this desk with a melting point of 130°F. And suppose that
one Marine had with him a tiny capsule containing a seed of ice-nine, a new
way for the atoms of water to stack and lock, to freeze. If that Marine threw that
seed into the nearest puddle…?”
“The puddle would freeze?” I guessed.
“And all the muck around the puddle?”
“It would freeze.”
“And all the puddles in the frozen muck? They would freeze?”
“You bet they would, and the United States Marines would rise from the swamp
and march on.”

“There is such stuff?”
“No, no, no, no ... if you'd been listening to what I have been trying to tell you
about pure research men, you wouldn't ask such a question. Pure research
men work on what fascinates them, not on what fascinates other people.”
“If the streams flowing through the swamp froze as ice-nine, what about the
rivers and lakes the streams fed?”
“They'd freeze.”
“And the oceans the frozen rivers fed?"
“They'd freeze, of course.”
“And the springs feeding the frozen lakes and streams, and all the water
underground feeding the springs?”
“They'd freeze, damn it!”
“And the rain?”
“When it fell, it would freeze into hard little hobnails of ice-nine and that would
be the end of the world.”
E.R. Lippincott et al., Science 164, p. 1482 (1969)
Infrared and Raman spectra completely different from
normal water
K. Morokuma, Chem. Phys. Lett. 4, p. 358 (1969)
L.C. Allen, P.A. Kollman, Science 167, p.1443 (March 1970)
“Explained” properties by quantum chemistry
(semi-empirical CNDO calculations)
L.C. Allen, P.A. Kollman, J. Am. Chem. Soc. 92, p. 4108 (July 1970)
Presented evidence that polywater does not exist
(ab initio calculations)
Special properties caused by solution of various impurities from capillary walls
What could be learned?
Role of media: popular journals, newspapers
(fear of ice-nine)
What is “New York Times physics”?
Sociology of scientist world
How efficient the scientific method can be (L.C. Allen)
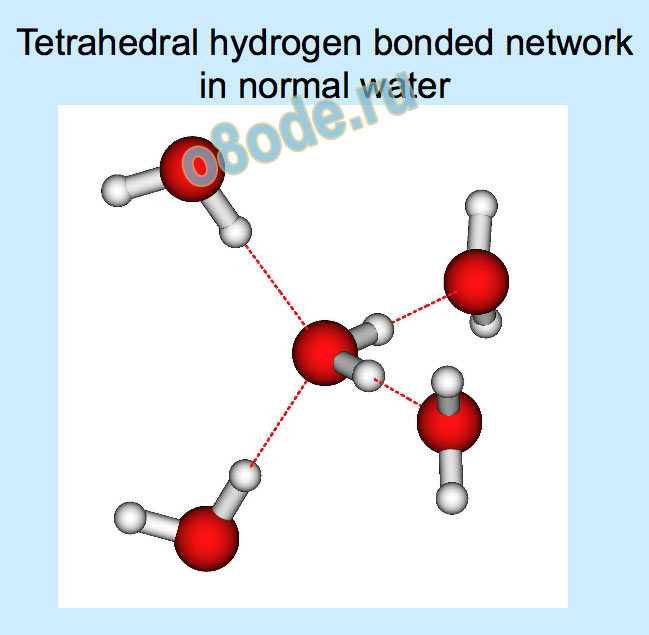
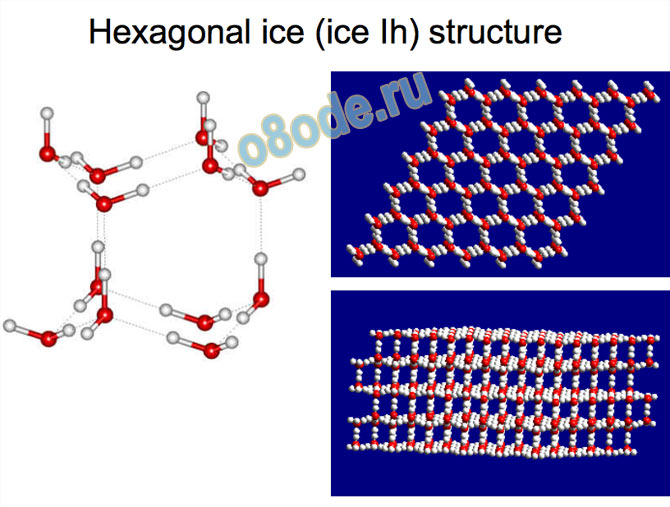
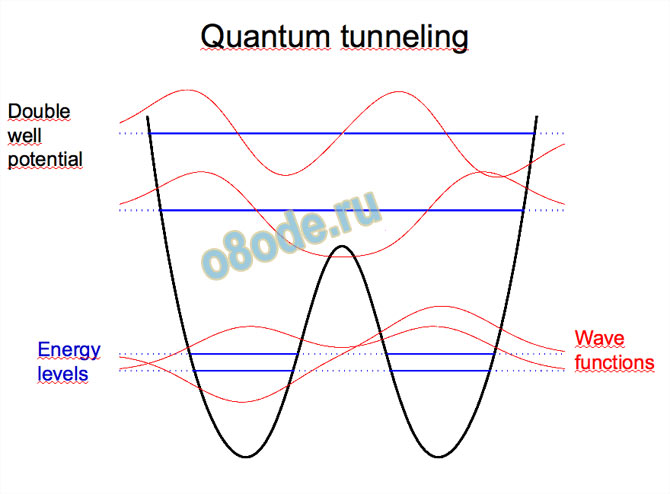
Structure, motion, and properties of water are determined by energy landscape (potential surface, force field, forces between the molecules)
First ab initio calculated water potentials:
Popkie, Kistenmacher, Clementi (IBM)
J. Chem. Phys. 59, 1325 (1973)
Hartree-Fock calculations, 190 points
Matsuoka, Clementi, Yoshimine (IBM)
J. Chem. Phys. 64,1351 (1976)
Configuration-Interaction calculations, 66 points
Based on > 2500 CCSD(T) points
extrapolated to CBS limit at MP2 level
Analytic representation, correct long-range behavior
Polarizable, includes many-body polarization effects
Supplemented with 3-body exchange terms
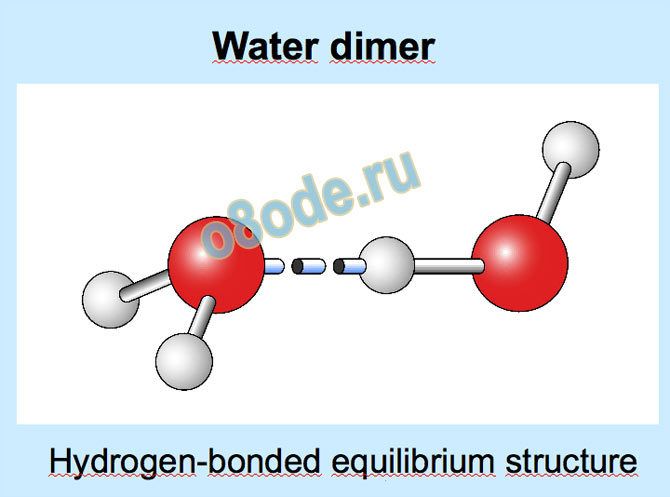
Tested by spectroscopy on dimer and trimer
Used in MD simulations for liquid water