Protonated Water Clusters
Протонированные кластеры воды
K. D. Jordan Department of Chemistry University of Pittsburgh Pittsburgh, PA Proton Solvation and Transport Workshop , June 25, 2006
Magic number (H2O)21+ cluster
Support National Science Foundation Department of Energy

Co-workers
R. A. Christie, J. Cui, E. M. Myshakin, T. H. Choi - Univ. Pittsburgh M. A. Johnson, J. -W. Shin, N. I. Hammer, E. G. Diken - Yale M. A. Duncan, R. S. Walters, T. D. Jaeger - Univ. of Georgia
Computational resources
• CMMS (Univ. of Pittsburgh) • Pittsburgh Supercomputing
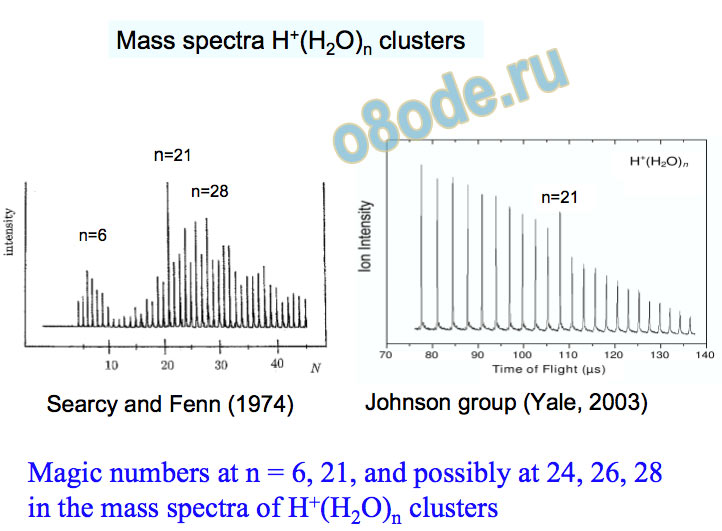
Center • Maui HPCC Johnson group (Yale, 2003) Searcy and Fenn (1974) Magic numbers at n = 6, 21, and possibly at 24, 26, 28 in the mass spectra of H+(H2O)n clusters n=6 n=21 n=28 n=21
Mass spectra H+(H2O)n clusters n = 2-30
hv Vibrational predissociation spectroscopy (Johnson and Duncan groups)
• Greatly increased sensitivity over simple absorption spectroscopy
• Ar-tagged clusters tend to be appreciably colder (T ≈ 140 K), yielding simpler spectra
H+(H2O)n H+(H2O)n-1 + H2O hv H+(H2O)n + Ar H+(H2O)nAr n = 1-15
Last few years – the combination of experiments and theory has led to significant advances in our understanding of the properties of protonated water clusters

1. Shin, Hammer, Diken, Johnson, Walters, Jaeger, Duncan, Christie, Jordan: Science 2004.
2. Miyazaki, Fujii, Ebatu, Mikami: Science 2004.
3. Wu, Lin, Chang, Jiang, Kuo, Klein: JCP 2005.
4. Lin, Wu, Wang, Lee, Chang, Kuo, Klein, PCCP 2005.
5. Lisy, unpublished results.
Theoretical studies
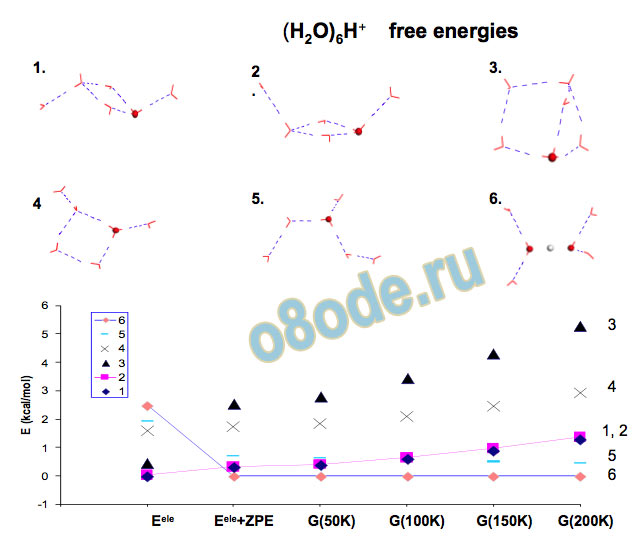
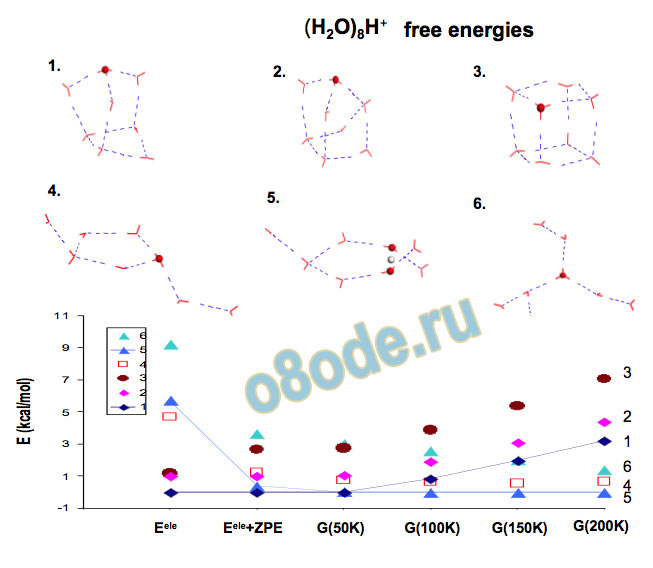
Parallel tempering Monte Carlio simulations with the MSEVB potential of Voth and co-workers to identify low energy minima DFT (Becke3LYP) and MP2 ab initio calculations of low energy local minima and of vibrational spectra
Recently we have examined the use of the tight-binding DFT for describing the potential energy landscapes of protonated water clusters
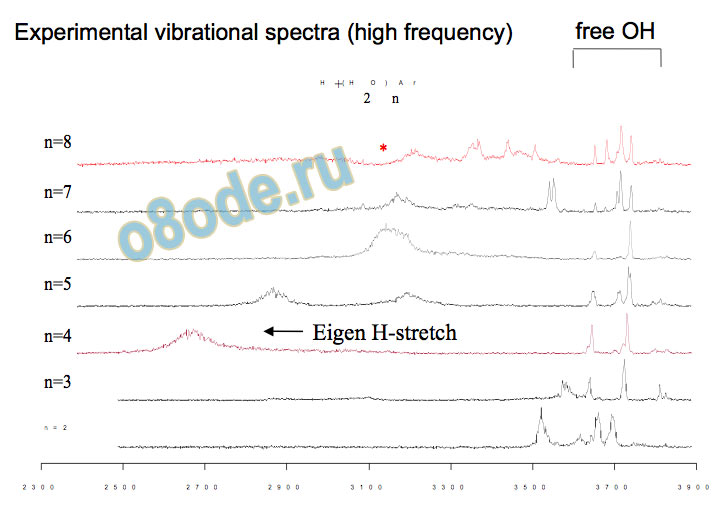
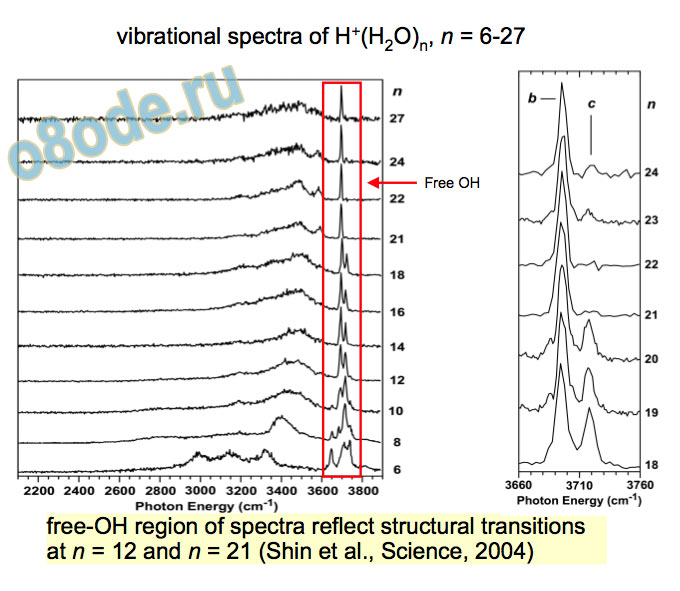
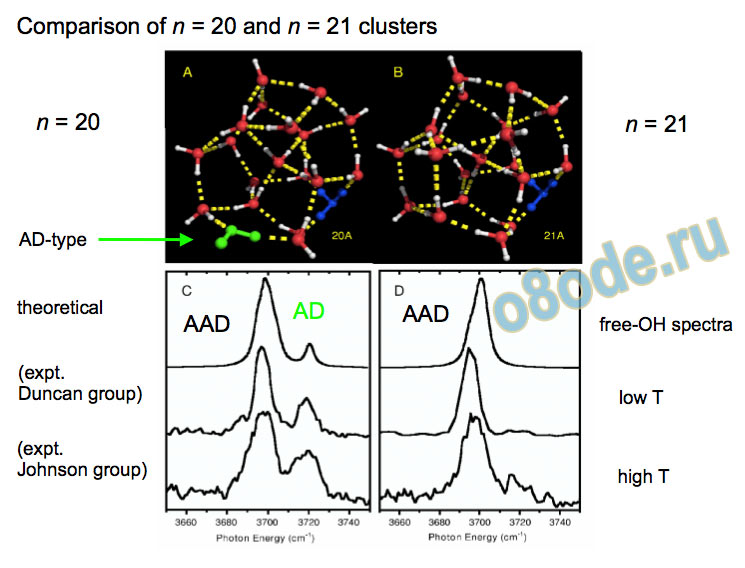
Experimental vibrational spectra (high frequency) free OH Eigen H-stretch vibrational spectra of H+(H2O)n, n = 6-27 free-OH region of spectra reflect structural transitions at n = 12 and n = 21 (Shin et al., Science, 2004)
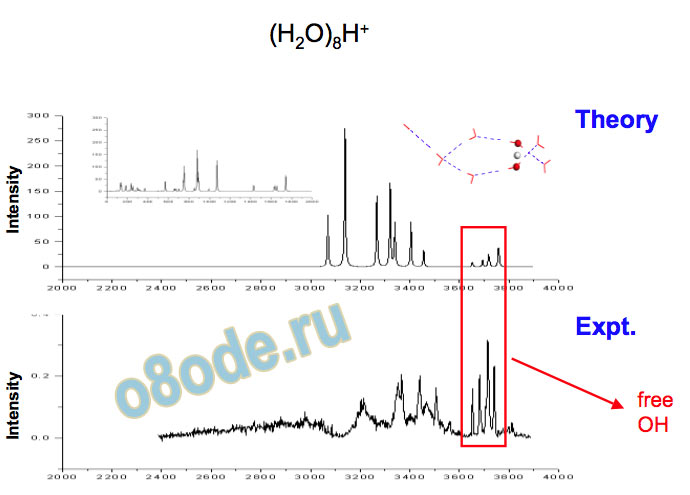
Free OH a and d are the asymmetric and symmetric free-OH stretches of dangling acceptor-only (type A) H2O molecules
Different types of free OH vibrations - example: H+(H2O)8 b is free-OH stretch of an acceptor-acceptor-donor (type AAD) H2O c is free-OH stretch of an acceptor-donor (type AD) H2O
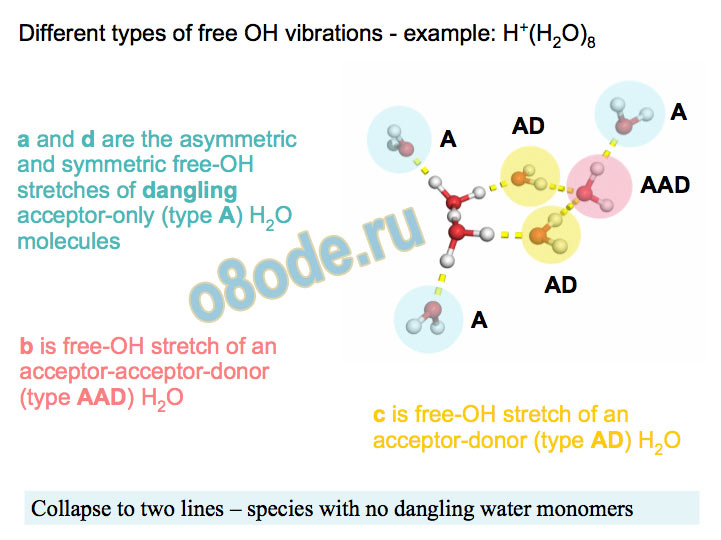
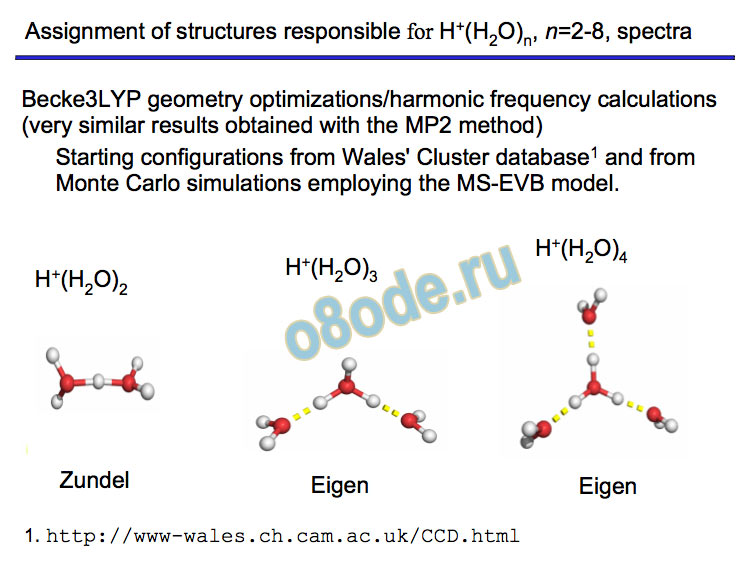
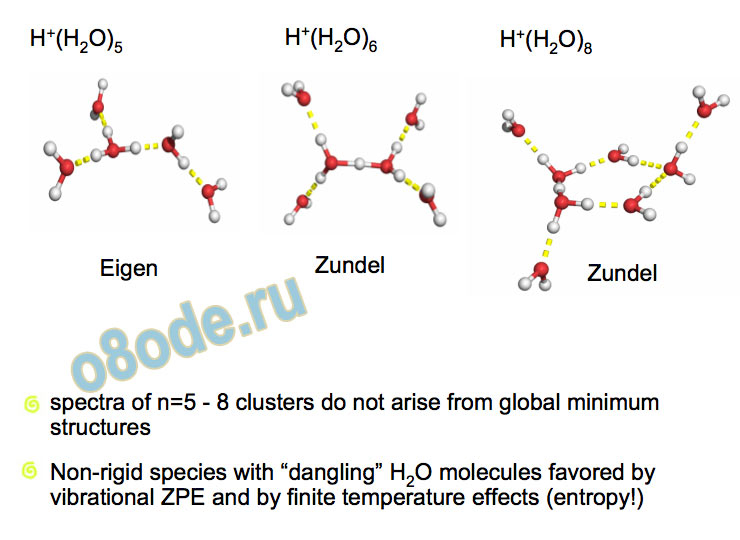
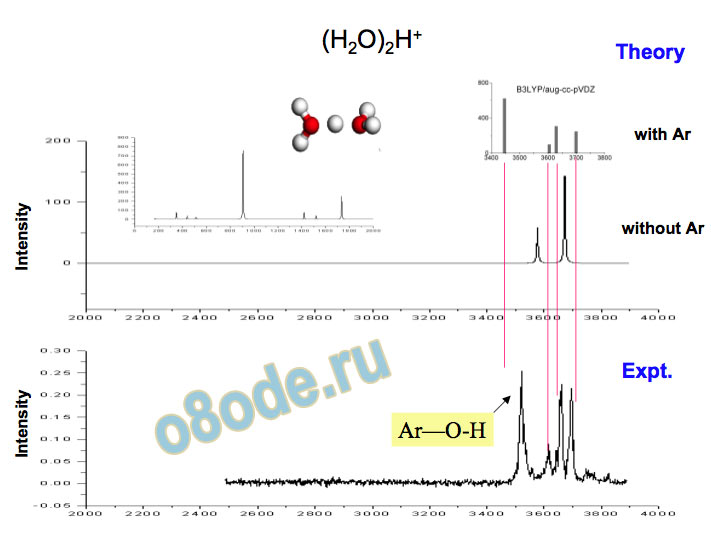
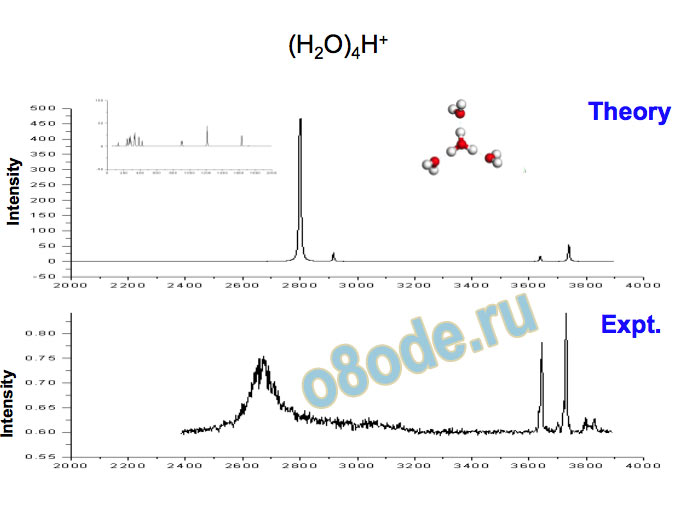
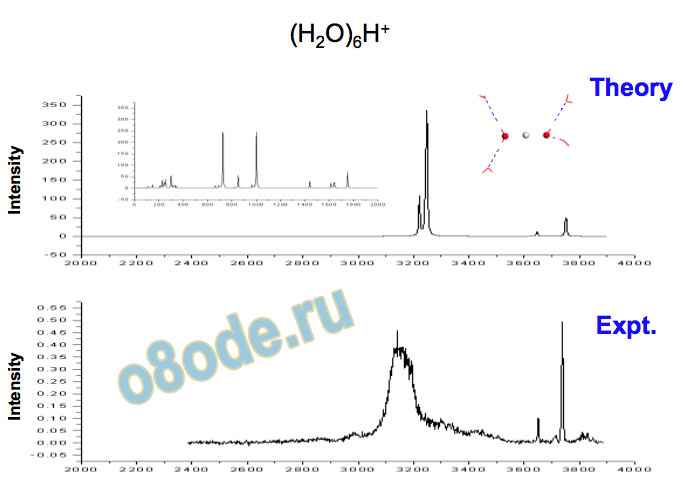
Collapse to two lines – species with no dangling water monomers Assignment of structures responsible for H+(H2O)n, n=2-8, spectra Becke3LYP geometry optimizations/harmonic frequency calculations (very similar results obtained with the MP2 method) Starting configurations from Wales' Cluster database1 and from Monte Carlo simulations employing the MS-EVB model.
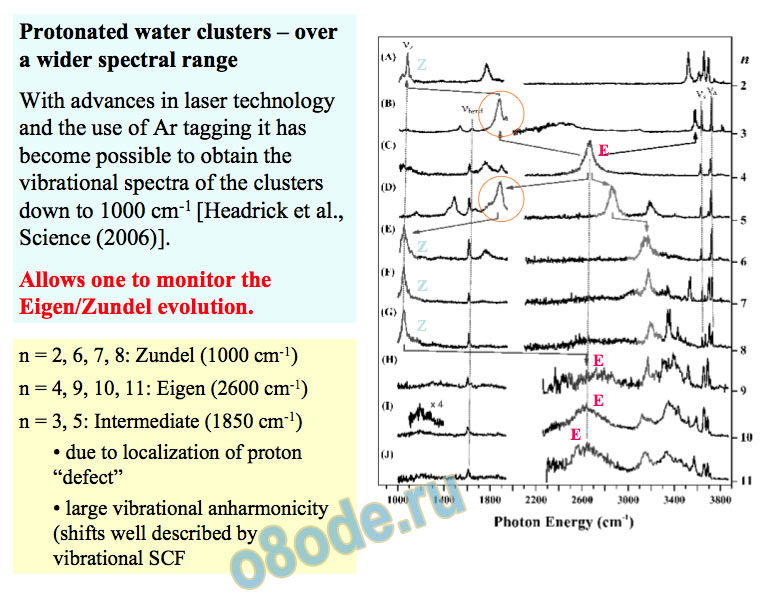
Protonated water clusters – over a wider spectral range With advances in laser technology and the use of Ar tagging it has become possible to obtain the vibrational spectra of the clusters down to 1000 cm-1 [Headrick et al., Science (2006)].
Allows one to monitor the Eigen/Zundel evolution.
n = 2, 6, 7, 8: Zundel (1000 cm-1) n = 4, 9, 10, 11: Eigen (2600 cm-1) n = 3, 5: Intermediate (1850 cm-1)
• due to localization of proton “defect”
• large vibrational anharmonicity (shifts well described by vibrational SCF
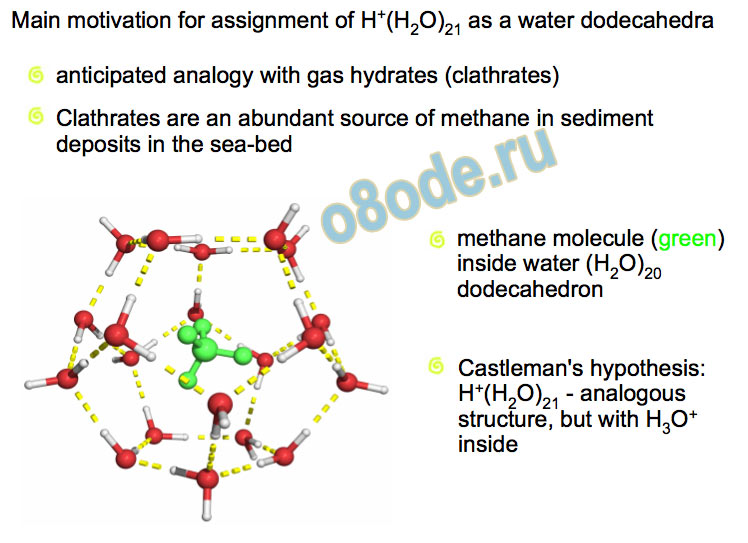
Main motivation for assignment of H+(H2O)21 as a water dodecahedra methane molecule (green)
Clathrates are an abundant source of methane in sediment deposits in the sea-bed Castleman's hypothesis:
H+(H2O)21 - analogous structure, but with H3O+
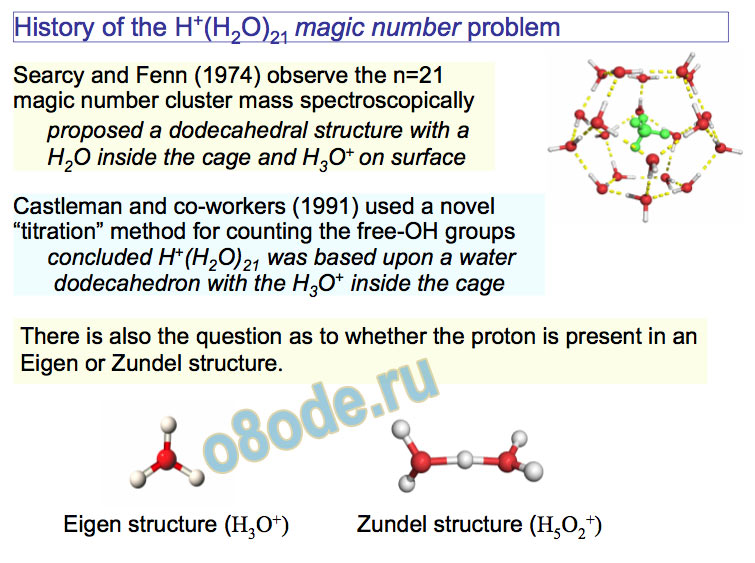
inside anticipated analogy with gas hydrates (clathrates) History of the H+(H2O)21 magic number problem Castleman and co-workers (1991) used a novel “titration” method for counting the free-OH groups concluded H+(H2O)21 was based upon a water dodecahedron with the H3O+ inside the cage Searcy and Fenn (1974) observe the n=21 magic number cluster mass spectroscopically proposed a dodecahedral structure with a H2O inside the cage and H3O+ on surface There is also the question as to whether the proton is present in an Eigen or Zundel structure.
Eigen structure (H3O+) Zundel structure (H5O2+)
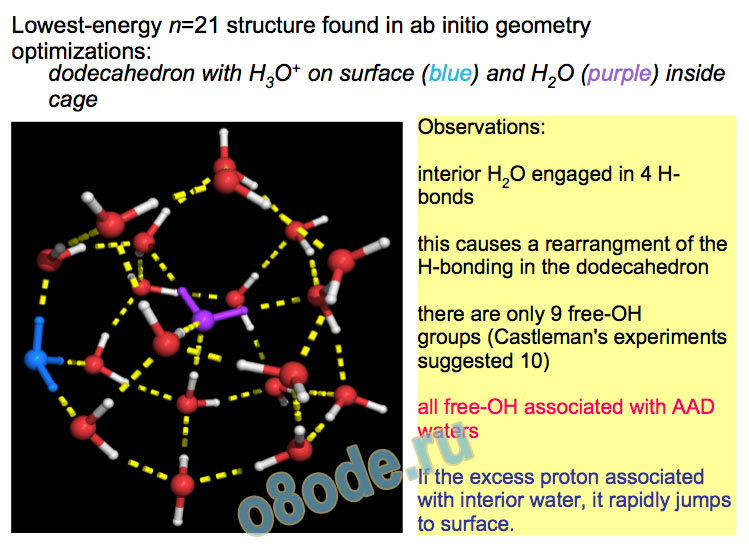
Lowest-energy n=21 structure found in ab initio geometry optimizations:
Comparison of n = 20 and n = 21 clusters n = 20 n = 21
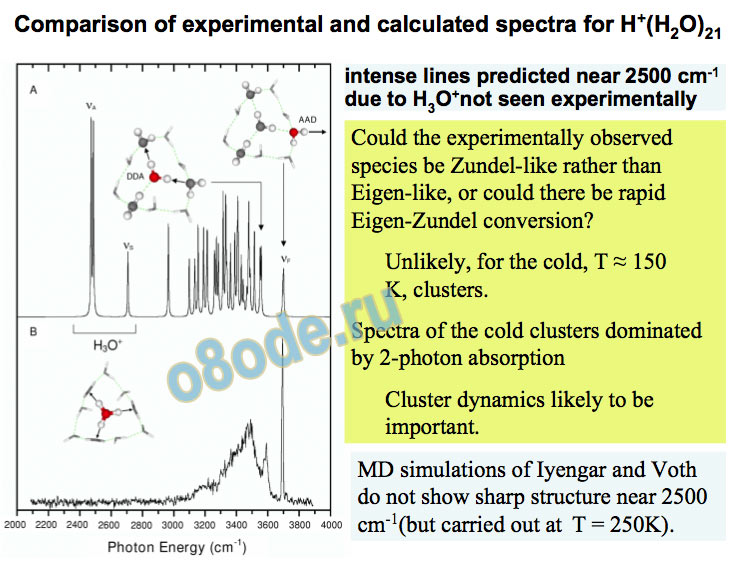
Comparison of experimental and calculated spectra for H+(H2O)21 intense lines predicted near 2500 cm-1 due to H3O+not seen experimentally Could the experimentally observed species be Zundel-like rather than Eigen-like, or could there be rapid Eigen-Zundel conversion?
Unlikely, for the cold, T ≈ 150 K, clusters.
Spectra of the cold clusters dominated by 2-photon absorption Cluster dynamics likely to be important.
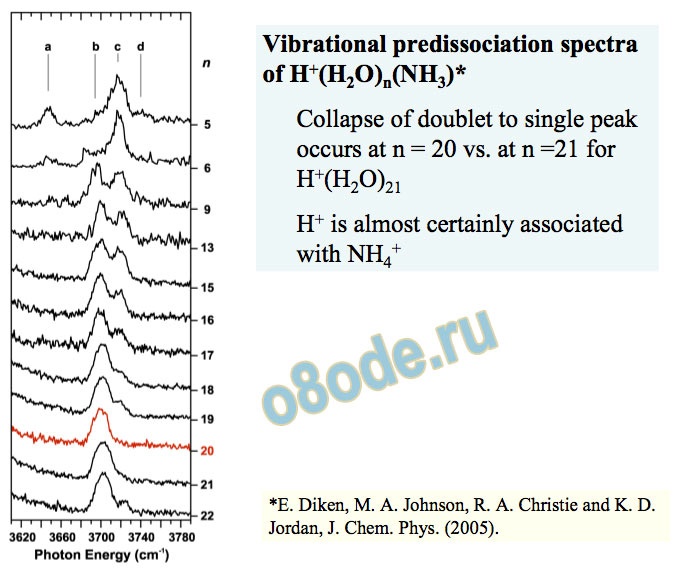
MD simulations of Iyengar and Voth do not show sharp structure near 2500 cm-1(but carried out at T = 250K). Vibrational predissociation spectra of H+(H2O)n(NH3)
Collapse of doublet to single peak occurs at n = 20 vs. at n =21 for H+(H2O)21 H+ is almost certainly associated with NH4+
Again, theory favors
E. Diken, M. A. Johnson, R. A. Christie and K. D. Jordan, J. Chem. Phys. (2005).
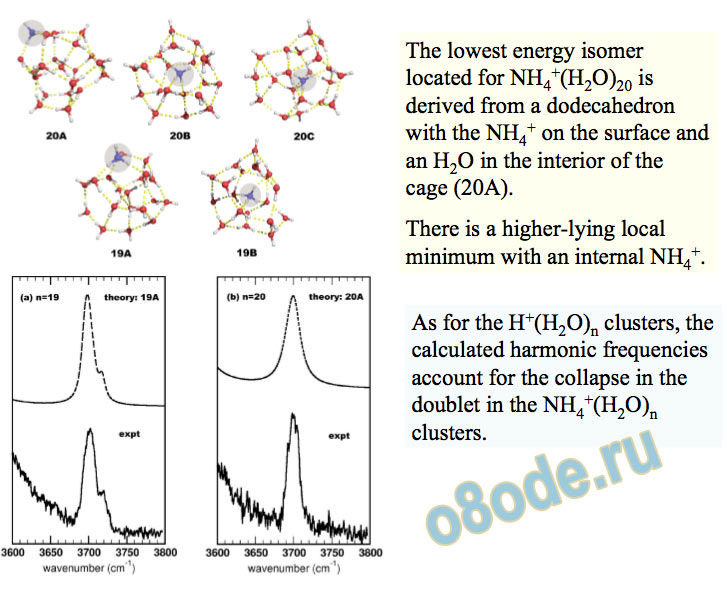
The lowest energy isomer located for NH4+(H2O)20 is derived from a dodecahedron with the NH4+ on the surface and an H2O in the interior of the cage (20A).
There is a higher-lying local minimum with an internal NH4+.
As for the H+(H2O)n clusters, the calculated harmonic frequencies account for the collapse in the doublet in the NH4+(H2O)n clusters.
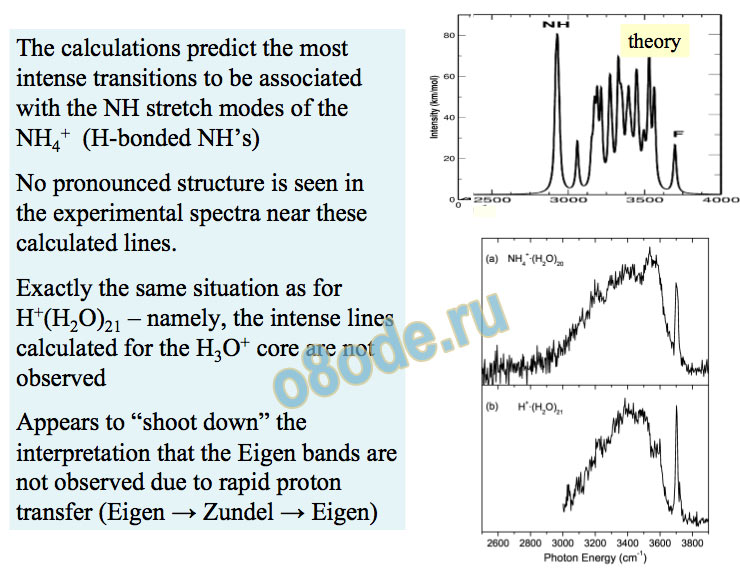
The calculations predict the most intense transitions to be associated with the NH stretch modes of the NH4+ (H-bonded NH’s) No pronounced structure is seen in the experimental spectra near these calculated lines.
Exactly the same situation as for H+(H2O)21 – namely, the intense lines calculated for the H3O+ core are not observed
Appears to “shoot down” the interpretation that the Eigen bands are not observed due to rapid proton transfer (Eigen → Zundel → Eigen) theory
Magic number clusters: starting T estimated to be ≈ 140K. (based on comparison of the mass spectra to those of Bondeby et al.) Absorption of a single OH stretch photon not expected to give H2O evaporation on the timescale of the expt. (10-5 sec.) In fact, the vibrational predissociation spectra of H+(H2O)21 (Johnson + Duncan groups) and NH4+(H2O)20 (Johnson group) are multiphoton.

Could the geometrical distortions resulting from absorption of a photon by the H3O+ or NH4+ result in sufficiently large frequency shifts so as to turn off absorption of subsequent photons.
This idea can be tested in a two laser experiment Estimated melting T ~ 165 K (from simulations)
Cluster initially solid-like
