Titanium electrode in Ionization Alkaline process
Hello Oleg,
My name Michael. I am interesting in Titanium electrode in Ionization Alkaline process. What do you think about this and different kind of metal for electrodes.
I will be waiting for your reply
Thank you, Michael
Dear Michael,
Thank you very much for your letter and your interest in our site-page.
As far as I know the ionized water is the product of mild electrolysis which takes place in the ionized water unit. So ionized water is treated tap water that has not only been filtered, but has also been reformed in that it provides reduced water with a large mass of electrons that can be donated to active oxygen in the body to block the oxidation of normal cells.
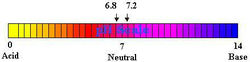
It is a well known fact that normal tap water with a pH of 7 is approximately neutral on the pH scale of 0 to 14. When measured with an oxidation potential meter its redox potential is approximately +400 to +500 mV. Because it has a positive redox potential, it is apt to acquire electrons and oxidize other molecules. Reduced Ionized Water, in contrast to the ordinary water, has a negative redox potential of approximately -250 to -350 mV. This means it has a large mass of electrons ready to donate to electron active oxygen.
The Ionized Water unit, slightly taller and thicker than a large dictionary on end, is an electrical appliance connected to your kitchen water supply to perform electrolysis on tap water before you drink it or use it in the kitchen for cooking or cleaning.
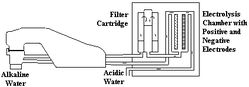
The scheme of water-ionizer (the picture was taken from site-page www.ionizers.org/water.html)
While the ionizer is working cations, positive ions, gather at the negative electrodes to create cathodic water (reduced water). Anions, negatively charged ions, gather at the positive electrode to make anodic water (oxidized water).

Sincerely yours,
Dr. Oleg V. Mosin